YOUTH ATHLETE DEVELOPMENT AND NUTRITION
Published
August 2021
Author
Ben Desbrow, PhD

KEY POINTS
- Adolescence is a period of natural experimentation, and is particularly important in terms of establishing the connection between diet, exercise and body image.
- An adolescent’s peers become increasingly powerful moderators of all behaviors, including eating.
- The pathway to elite sports performance is complex, and rarely forecast by success at junior levels.
- Stakeholders involved in managing developing athletes have a responsibility to prioritize sound physical and mental development while integrating principles of sport nutrition success.
- The assessment of body composition in adolescent athletes is rarely needed, and should only be performed by a trained professional once the performance level and nutrition literacy skills are considered, and the athlete and guardian have consented.
- New data facilitates a more accurate estimation of basal energy requirements in adolescent athletes.
INTRODUCTION
Regular exercise provides many benefits to young people, including social interaction, improved physical health, and the development of self-identity and self-esteem. In addition, the second decade of life is an important time in establishing an individual’s relationship with food, and the lifelong connection between diet, exercise and body image. The pathway to elite adult performance is considered multifaceted and nonlinear (i.e., success at junior levels infrequently predicts elite adult performance). As such, athlete health remains of utmost importance during this period.
THE CHANGING FOCUS OF YOUTH ATHLETE DEVELOPMENT
Clearly, participation in sport plays an important role in supporting psychological well-being and developing a healthy self-image for most adolescents (Ekeland et al., 2005). However, for many years, increased rates of disturbed eating attitudes/behaviors and body dissatisfaction have been evident in sports emphasizing leanness (Martinsen et al., 2010). This evidence, combined with high-profile advocacy from former athletes (Cain, 2019) and a greater appreciation of the long-term health consequences of mismanaging the diet-exercise relationship in younger athletes (Mountjoy et al., 2014), has led to calls to fundamentally change how sports manage aspiring athletes (Ackerman et al., 2020). These changes may involve avoiding unwarranted nutrition- and food-related discussions, abolishing body composition and weight assessments, raising awareness of the negative effects of chronic low energy availability, and disrupting toxic training environments featuring abusive body shaming, including the use of training strategies designed to manipulate an athlete’s physique independent of performance. The framework recommends sports nutrition support decisions be guided by athlete performance level and the nutrition literacy skills of the recipent, and requires athlete and guardian consent when undertaking body composition assessments (Figure 1).
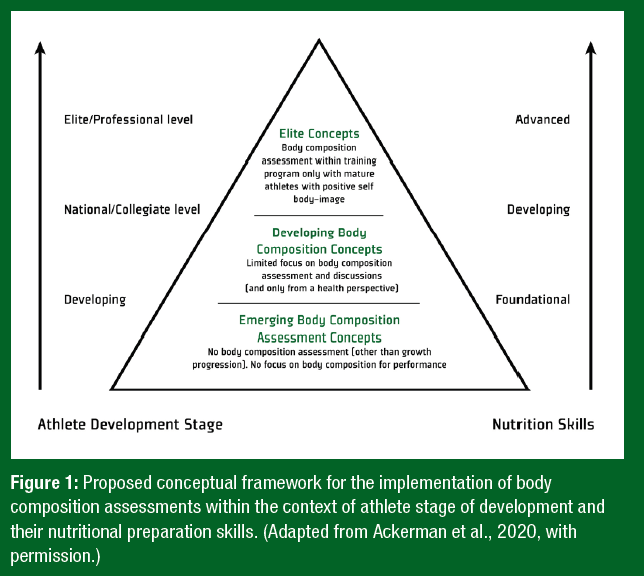
In response, some sporting organizations have attempted to safeguard their custodianship of adolescent athletes by publishing expected stakeholder behaviors. For example, in 2019 Gymnastics Australia released body positive guidelines providing specific recommendations on appropriate language, the frequency and delivery of nutrition education, and body composition assessment within gymnastic environments (Gymnastics Australia, 2019). The extent to which such strategies moderate the behavior of coaches, parents, and support staff and/or ultimately reduce the incidence of undesirable health outcomes in developing athletes has yet to be elucidated. In the interim, it seems prudent to suggest that those involved in adolescent sport acquire the knowledge and support to ensure that appropriate, evidence-based nutrition care is provided to developing athletes.
ENERGY NEEDS OF DEVELOPING ATHLETES
Throughout adolescence, adequate energy is required to meet both the growth and development needs of the individual as well as the substrate demands associated with general physical activity, training, and competition. Changes in training and competition load, participation in more than one competitive sport, part-time employment and/or concurrent compensatory sedentary behaviors may all impact energy needs. Determining the individual energy requirements of adolescent athletes is further complicated by metabolic and hormonal variability within and between individuals (Petrie et al., 2004), and methodological difficulties in estimating both energy intake and energy expenditure (Burke et al., 2001).
Growth during puberty is directly related to the hormonal changes that accompany sexual development. While girls generally start their growth spurt and attain peak height velocity two years earlier than boys (~12 yr vs. ~14 yr), other factors may also influence the timing of growth. This includes ethnicity, in which school-age individuals with European ancestry have around 6 months younger skeletal age than chronologically matched individuals with Asian or African heritage (Grgic et al., 2020). The energy needs for growth (a component of the energy requirements of adolescent athletes) consist of two parts: the energy expended to synthesize those tissues, and the energy deposited in growing tissues (Torun, 2005).
The energy expended to synthesize new tissues can be directly measured via the doubly labelled water (DLW) method or (more commonly) estimated indirectly via measures of resting metabolic rate (RMR). The use of adult-based equations to predict RMR in adolescent athletes is not recommended, as these have been shown to underestimate energy expenditure (up to 300 kcal/day) compared with indirect calorimetry measures (Loureiro et al., 2015). Recently, new predictive resting metabolism equations have been developed from a cohort of male and female junior athletes (n=126) who each undertook an indirect calorimetry assessment of RMR under standardized conditions. The cohort included athletes from a range of sports with an average age of 16.5 years (range, 13.1-19.7 yr) (Reale et al., 2020). The predictive RMR equations for developing athletes were:
RMR (kcal/day) = 11.1 × Body Mass (kg) + 8.4 × Height (cm) – (340 male or 537 female)
The energy deposited in growing tissues is more difficult to measure, but it is considered small and is commonly estimated as ~2.0 kcal/g of daily weight gain (e.g., for a 15-year-old male gaining 6 kg/year = ~33 kcal/day) (WHO, 1983). Hence, while two energy components of growth may alter total caloric requirements, evidence suggests that changes associated with physical activity and/or athletic training are likely to have a greater influence on total energy demands of adolescent athletes (Torun, 2005).
Energy expenditure in adolescent athletes can be accurately measured with methods such as DLW or indirect calorimetry. However, given these methods are expensive and rely on complex techniques, widely available methods for estimating individual energy expenditures warrant consideration. Wearable technologies incorporating accelerometers represent a relatively inexpensive alternative to estimate individual energy expenditure in younger populations. Recent reviews have summarized the validity of different “wearables” to estimate total energy expenditure and the energy cost of physical activity against DLW (Sardinha & Judice, 2017), and indirect calorimetry (Lynch et al., 2019) in younger (general) populations. These reviews suggest that (1) there is no currently ideal device, (2) accelerometers tend to underestimate energy expenditure during activities such as incline walking, bicycling and carrying items, and (3) more accurate results are recorded when the accelerometer is placed closer to the center of mass (e.g., hip compared with wrist or ankle) of the individual.
The accurate determination of energy intakes and expenditures is important, as it appears that low-energy availability (LEA) and potential symptoms of relative energy deficiency in sport (RED-S) in young individuals undertaking heavy training is common (Muia et al., 2016; Rogers et al., 2021). In developing athletes, LEA may lead to a number of serious health consequences, including delayed puberty, menstrual irregularities, poor bone health, short stature, the development of disordered eating behaviors and increased risk of injury (Desbrow et al., 2019). Furthermore, in females ≤14 years gynecological age, the effects of low EA may be more pronounced (Loucks, 2006). Conversely, some developing athletes (e.g., those who participate in throwing events) demonstrate anthropometric characteristics consistent with chronic disease risk (Hirsch et al., 2016). In this context, severe and prolonged energy restriction is not recommended, with weight maintenance, rather than weight loss, considered a more appropriate management strategy in developing individuals (Desbrow et al., 2014).
Macronutrient Needs of Developing Athletes
Adolescents require protein to support general growth and development, in addition to enhancing the response to exercise training (Witard et al., 2019). During peak growth, increases in lean body mass can reach ~2.3 g/day in females and ~3.8 g/day in males, which represents a ~3-fold increase from the prepubertal period (Forbes, 1964). In addition, longitudinal data indicate that physically active youth accrue greater increases in lean body mass than their sedentary peers (Baxter-Jones et al., 2008). While regular training does not appear to influence protein turnover in early adolescence (Boisseau et al., 2005), one proposed explanation for the enhanced lean mass deposition observed during puberty relates to enhanced anabolic sensitivity (i.e., a greater efficiency of dietary protein utilization) (Beckett et al., 1997). This notion has received further support by the demonstration that adolescents have greater whole-body net balance when provided with small to optimal amounts of post-exercise protein compared with weight-stable adults (Mazzulla et al., 2018). Furthermore, an increased efficiency of amino acid use would explain previous nitrogen-balance studies, which did not demonstrate additional dietary protein intakes were required in adolescent sprint athletes to maintain a positive nitrogen balance during their peak growth phase (Aerenhouts et al., 2013).
Total energy intake is an important consideration in the assessment of protein requirements. With suboptimal energy intake, endogenous protein is mobilized as well as liver glycogen to maintain homeostasis of blood glucose, potentially reducing the availability of protein for its primary functions. Provided energy intake is adequate, it appears that protein recommendations to maximize whole-body net balance after exercise are primarily influenced by total body and fat-free mass. Protein intake at ~0.11 g/kg/h during post-exercise recovery or the equivalent of ~1.5 g/kg/d (e.g., ~0.3 g protein/kg × 5 mealtimes) appears sufficient to replace any exercise-induced amino acid oxidative losses, enhance whole body net protein balance, and support the normal growth and development of adolescent athletes (Mazzulla et al., 2018).
The duration and intensity of exercise sessions determines carbohydrate (CHO) utilization patterns and refueling requirements. There is little evidence to suggest that the utilization of CHO in adolescents differs substantially from that of adults (for review, see Desbrow & Leveritt, 2020). Dietary CHO needs should be considered in light of the training loads and competition characteristics that are typically undertaken by adolescent athletes. These may differ from those of adult athletes in a number of ways. Firstly, developing athletes can be involved with numerous organizations (e.g., schools, clubs and regions), creating different competition frequencies and formats, such as sports carnivals, representative events and trials. It is also common for aspiring adolescent athletes to participate in a number of different sports. These different energy demands and subsequent CHO requirements must be considered, particularly when the participation in different sports is concurrent. While dietary strategies incorporating the use of refined CHO to manipulate metabolism during endurance sport (e.g., CHO loading and/or multiple transportable forms of CHO throughout exercise) are likely to improve performance in events of greater than 90 minutes’ duration (Burke et al., 2011), these recommendations should only be employed in relevant situations, which is less likely for adolescent athletes given the shorter duration of many events.
Adequate dietary fat intake is needed to meet requirements for fat-soluble vitamins and essential fatty acids, and helps provide energy to support growth and maturation. In addition, evidence suggests that maximal fat oxidation rates (relative to lean mass) are slightly higher in athletes younger than 18 years (Randell et al., 2017). To date, dietary strategies promoting the role of the intramuscular triacylglycerols on performance and the effect of training in a CHO-depleted state on adolescent endurance athletes remain unstudied. Since chronically high fat intakes are associated with increased chronic disease risk, the recommendation for type and total fat intake by adolescent athletes remains in accordance with public health guidelines. Typically, these guidelines suggest a dietary fat intake of 20–35% of total energy, with saturated/trans fatty acids providing no more than 10% of total energy intake (Department of Agriculture, 2020).
When energy demands change, sports nutrition recommendations encourage athletes to manipulate dietary intake to support daily performance and optimize adaptations to training, a concept known more commonly as “fuel for the work required” (Impey et al., 2018). Adolescent athletes are likely to require support to develop a “food first” approach to matching energy intakes with increased training loads and may benefit from practical resources translating changes in macronutrient needs to food selection (Figure 2).

Micronutrient Needs for Adolescent Athletes
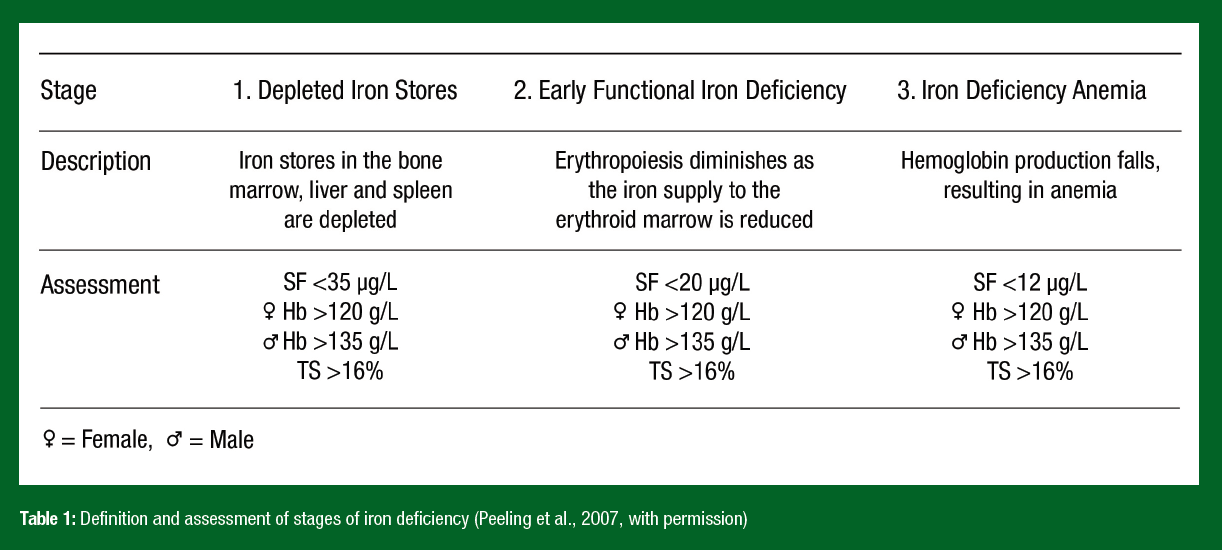
Depleted iron stores, without clinical symptoms, are observed frequently in studies conducted on adolescent athletes (particularly endurance competitors) (Shoemaker et al., 2020). Interpreting one-off measures of iron status markers (e.g., serum ferritin) in developing athletes should be done with caution for several reasons: (1) cut-off values for ferritin are not standardized in studies of young athletes, (2) athletes generally have lower levels of ferritin than non-athletes, (3) sex differences are evident between males and females during adolescence, and (4) ferritin levels can be falsely positive with mild infection, injury or physiological stress (Peeling & Deakin, 2021). Detection and early treatment of iron depletion in adolescent athletes is warranted. This is because growth increases iron requirements in adolescents compared with older athletes, resulting in the progression from low iron stores to a state of rapid iron deficiency. Reference values (Peeling et al., 2007) and strategies to address poor iron status in athletes (AIS, 2019) have been established (Table 1).
In terms of performance, even mild tissue decrements in iron have the potential to adversely affect endurance capacity and aerobic adaptation to training (Rodenberg & Gustafson, 2007). Indeed, a recent study involving over 70 adolescent female athletes indicated a moderate relationship between athletic performance, the concentration of soluble transferrin receptors, and dietary iron intake, emphasizing the importance of iron intake for aspiring young female athletes (Shoemaker et al., 2019). In adolescent female endurance athletes, suboptimal iron status is attributed to a combination of low iron intake, low iron bioavailability, and high requirements associated with training and blood loss (e.g., increased red cell mass, menstruation, hematuria, hemolysis) (Koehler et al., 2012). In contrast, suboptimal iron status in adolescent male athletes is associated more with high physiological requirements (i.e., training and growth) than with diet.
Recommendations for iron for developing girls accounts for iron lost from menstruation. Often a population reference value is used as an age cut-off for menarche (e.g., 14 yr). Individual recommendations should be adjusted when individual differences exist (particularly when menarche occurs earlier). It is also possible that vegetarian athletes have increased requirements due to low iron bioavailability of non-heme iron sources. However, scientific advisory summaries indicate that when comparing vegetarians with non-vegetarians, most studies demonstrated no significant differences in dietary iron intake or hemoglobin concentrations. Although serum ferritin concentrations are statistically significantly lower in vegetarians on a consistent basis, they are usually within the reference ranges (SCAN, 2010). That said, it seems prudent to ensure young vegetarian athletes monitor iron status routinely.
Calcium requirements are highest during the pubertal growth spurt. The rate of skeletal calcium accretion during adolescence is estimated to be around 300 mg/d. There are currently no specific recommendations for calcium intake for athletes, so until further studies are undertaken, population reference standards can be used as a benchmark for assessing adequacy. Calcium recommendations are based on estimates of urinary and sweat losses and assume a net calcium absorption from food (often ~25–35%). Recommendations for adolescents vary between regions, with values ranging from 800 mg/day (e.g.. UK females, age 15-18 yr) to 1300 mg/day (e.g., in US, Canada, and Australia for males and females, age 14–18 yr). High-intensity weight-bearing exercise, and, to some extent, resistance exercise increase bone mineral content in exercising adolescents (Elhakeem et al., 2020). While this effect is small (typically <6% difference) and unlikely to increase calcium requirements, enhanced bone mineral content may maximize peak hip strength and prevent osteoporosis in later life.
While vitamin D is best known for its role in bone health, it has many functions in other physiological systems (e.g., immune system, muscular system). Vitamin D insufficiency is also linked to skeletal muscle function, muscle pain and weakness, and inflammation and may potentially increase susceptibility to injury and slow the rate of rehabilitation from injury (for review, see de la Puente Yague et al., 2020). Hence, vitamin D status (particularly in adult athletes) has received considerable recent scientific attention. Currently, the influence of vitamin D status, and benefits of supplementation in young athletes identified as deficient, remains largely unknown. However, recent prospective studies suggest little correlation between serum levels of 25-hydroxyvitamin D and sports performance in younger athletes (Orysiak et al., 2018), even when vitamin D deficiencies are corrected (Bezuglov et al., 2019).
Similar to adult athletes, developing athletes are at high risk of vitamin D deficiency if they experience limited sun exposure (e.g., reside in latitudes above the 35th parallel, spend long periods training indoors, have dark skin, use sunscreen, or wear protective clothing). Typically, fixed amounts of vitamin D are recommended beyond infancy, until values for older adults are further increased to account for the reduced capacity of the skin to produce vitamin D with aging. Recommendations for vitamin D differ by region (Australia = 5 mg/day, US/Canada = 15 mg/day, European countries range from 10 to 20 mg/day). All authorities agree, however, that monitoring of 25-hydroxyvitamin D is important for at-risk groups.
FLUID NEEDS OF DEVELOPING ATHLETES
Young individuals appear to have the same capacity as adults to deal with thermal loads and exercise-tolerance time during exercise in the heat. However, the mechanisms by which young individuals dissipate heat loads during exercise differ from those of adults (Falk & Dotan, 2008). Children and adolescents appear to rely more on peripheral blood redistribution (radiative and conductive cooling) rather than sweating (evaporative cooling) to maintain thermal equilibrium (Barnes et al., 2019; Falk & Dotan, 2008). There is also evidence that adolescents who undertake regular training adapt by enhanced peripheral vasodilation (Roche et al., 2010), which is likely to improve non-evaporative cooling. While the timing of the transition from child-like to adult-like thermoregulatory mechanisms is likely to be related to pubertal development, it appears that these changes do not become physiologically evident until puberty has been completed (Falk et al., 1992).
There is some evidence suggesting an increased prevalence of heat illness associated with sport and activity in younger athletes (CDC, 2011). Heat illness may be influenced by poor hydration status along with other factors, such as undue physical exertion, insufficient cooling between exercise bouts, and inappropriate choices of clothing, including uniforms. Unfortunately, there is no evidence to determine the extent to which, if at all, fluid intake may modulate the risk of heat illness in adolescent athletes. This is because fluid monitoring studies on children and adolescents at risk of heat illness are scarce and often fail to report participants who actually experience heat illnesses. In contrast, field studies (Aragon-Vargas et al., 2013) and large cohort investigations (Barnes et al., 2019) indicate that trained adolescent athletes can experience significant deficits in fluid loss (>4% body mass) and high sweat rates (≤2.16 L/h) in response to exercise. Fluid shifts of this magnitude have the potential to induce signs/symptoms of hypohydration and affect exercise performance. As such, it appears prudent to apply the same fluid intake guidelines indicated for adult athletes. In addition, developing athletes may not recognize the signs or symptoms of heat stress, forget to drink unless reminded and continue to exercise to keep up with their peers. It is important that when environmental and contextual factors (e.g., intensity/duration of exercise, clothing, fluid availability) combine to increase thermoregulatory risks, that strategies to moderate metabolic heat loads in younger athletes be implemented proactively.
DIETARY SUPPLEMENTS AND ERGOGENIC AID USE BY DEVELOPING ATHLETES
The judicious use of specific dietary supplements and nutritional ergogenic aids may improve sporting performance in adults. However, their effectiveness and potential long-term effects have not been rigorously studied in younger populations, largely due to the ethical concept of beneficence (i.e., cost versus benefit). Despite this lack of scientific evidence, reported supplement use with the intent to improve sports performance among young athletes is common. The prevalence of supplement use among US children and adolescents (<18 yr) as a performance enhancer was 1.6% (Evans et al., 2012). Vitamin/mineral supplements, sports/protein powders, vitamin waters, creatine and caffeine are commonly identified as popular supplements used by adolescent athletes. Adolescent athletes take performance-enhancing dietary supplements for several reasons, such as pressure to achieve results, the pursuit of physical ideals, and peer, social and marketing pressures. In addition, some young elite athletes consider supplements critical for certain training adaptations, such as strength gains (Bloodworth et al., 2012).
In general, it is considered inappropriate for young athletes to be encouraged to consume dietary supplements for performance enhancement. This view is consistent with those of leading expert groups (Peeling et al., 2019). This recommendation excludes the clinical use of dietary supplements (e.g., calcium, iron, vitamin D) when taken under appropriate guidance from suitably qualified health professionals (e.g., a medical practitioner, sports dietitian). Apart from issues related to safety, the use of supplements in developing athletes overemphasizes their ability to manipulate performance. Younger populations have the potential for greater performance enhancement through maturation and experience in their sport, along with adherence to proper training, nutrition and rest regimens. It can also be argued that discouraging the use of dietary supplements downplays the win-at-all-costs mentality and sets an important example for young athletes.
PRACTICAL RECOMMENDATIONS
- Don’t be critical. Support adolescents to be right without making them wrong.
- Give a little nudge, and nudges are not mandates (e.g., cutting/peeling fruit and leaving it in a convenient location is a nudge, banning junk food is not).
- Don’t rely on rationale thought. Logic makes individuals think; passion makes individuals act. Find an emotive/fun approach and give a logical one too (e.g., cooking/recipe/tasting competitions).
- An adolescent’s peers become increasingly powerful moderators of all behaviors, including eating. Consider talking to and engaging peers, not parents.
- Adolescent experiences are not the same, and reasons underpinning individual behaviors vary considerably. Don’t make assumptions; instead, listen.
- “Silver bullets” are rarely found. Environments that support repetition and consistency work best.
SUMMARY
Adolescence is a period of significant growth and physical development that includes changes in body composition, metabolic and hormonal fluctuations, maturation of organ systems, and establishment of nutrient deposits, all of which may affect future health. Young athletes have unique nutritional requirements as a consequence of undertaking daily training and competition throughout this developmental period. To ensure an adolescent athlete fulfills their potential, it is important that stakeholders involved in managing younger athletes advocate for eating patterns that prioritize sound physical and mental development, while integrating the principles of sport nutrition success. The responsibility for the provision of appropriate nutrition care to developing adolescent athletes is shared among sporting organizations, coaches, parents, teachers and the athletes themselves.
The views expressed are those of the authors and do not necessarily reflect the position or policy of PepsiCo, Inc.
References:
Ackerman, K.E., T. Stellingwerff, K.J. Elliott-Sale, A. Baltzell, M. Cain, K. Goucher, L. Fleshman, and M.L. Mountjoy (2020). REDS (Relative Energy Deficiency in Sport): time for a revolution in sports culture and systems to improve athlete health and performance. Br. J. Sports Med. 54:369-370.
Aerenhouts, D., J. Van Cauwenberg, J.R. Poortmans, R. Hauspie, and P. Clarys (2013). Influence of growth rate on nitrogen balance in adolescent sprint athletes. Int. J. Sport Nutr. Exerc. Metab. 23:409-417.
AIS (2019). Austalian Institute of Sports. Sports Supplement Framework: Iron Supplement.
Aragon-Vargas, L., B. Wilk, B. Timmons, and O. Bar-Or (2013). Body weight changes in child and adolescent athletes during a triathlon competition. Eur. J. Appl. Physiol. 104:831-837.
Barnes, K.A., M.L. Anderson, J.R. Stofan, K.J. Dalrymple, A.J. Reimel, T.J. Roberts, R.K. Randell, C.T. Ungaro, and L.B. Baker (2019). Normative data for sweating rate, sweat sodium concentration, and sweat sodium loss in athletes: An update and analysis by sport. J. Sports Sci. 37:2356-2366.
Baxter-Jones, A.D., J.C. Eisenmann, R.L. Mirwald, R.A. Faulkner, and D.A. Bailey (2008). The influence of physical activity on lean mass accrual during adolescence: a longitudinal analysis. J. Appl. Physiol. 105:734-741.
Beckett, P.R., F. Jahoor, and K.C. Copeland (1997). The efficiency of dietary protein utilization is increased during puberty. J. Clin. Endocrinol. Metab. 82:2445-2449.
Bezuglov, E., A. Tikhonova, A. Zueva, V. Khaitin, A. Lyubushkina, E. Achkasov, Z. Waskiewicz, D. Gerasimuk, A. Zebrowska, P.T. Nikolaidis, T. Rosemann, and B. Knechtle (2019). The dependence of running speed and muscle strength on the serum concentration of vitamin D in young male professional football players residing in the Russian Federation. Nutrients 11:2405.
Bloodworth, A., A. Petroczi, R. Bailey, G. Pearce, and M. McNamee (2012). Doping and supplementation: the attitudes of talented young athletes. Scand. J. Med. Sci. Sports 22:293-301.
Boisseau, N., C. Persaud, A.A. Jackson, and J.R. Poortmans (2005). Training does not affect protein turnover in pre- and early pubertal female gymnasts. Eur. J. Appl. Physiol. 94:262-267.
Burke, L.M., G.R. Cox, N.K. Culmmings, and B. Desbrow (2001). Guidelines for daily carbohydrate intake: do athletes achieve them? Sports Med. 31:267-299.
Burke, L.M., J.A. Hawley, S.H. Wong, and A.E. Jeukendrup (2011). Carbohydrates for training and competition. J. Sports Sci. 29(Suppl 1):S17-S27.
Cain, M. (2019). I was the fastest girl in America, until I joined Nike. NY Times.
CDC (2011). Centers for Disease Control Prevention: Nonfatal sports and recreation heat illness treated in hospital emergency departments--United States, 2001-2009. MMWR Morb Mortal Wkly Rep, 60:977-980.
de la Puente Yague, M., L. Collado Yurrita, M.J. Ciudad Cabanas, and M.A. Cuadrado Cenzual (2020). Role of vitamin D in athletes and their performance: Current concepts and new trends. Nutrients 12:579.
Department of Agriculture & Department of Health and Human Services (2020). Dietary Guidelines for Americans, 2020-2025. Washington, D.C.
Desbrow, B., N.A. Burd, M. Tarnopolsky, D.R. Moore, and K.J. Elliott-Sale (2019). Nutrition for special populations: Young, female, and masters athletes. Int. J. Sport Nutr. Exerc. Metab. 29:220-227.
Desbrow, B., and M. Leveritt (2020). Nutritional issues for young athletes: children and adolescents. In: L. M. Burke, V. Deakin, & M. Minehan (Eds.), Clinical Sports Nutrition (6th ed): McGraw Hill.
Desbrow, B., J. McCormack, L.M. Burke, G.R. Cox, K. Fallon, M. Hislop, R. Logan, N. Marino, S.M. Sawyer, G. Shaw, A. Star, H. Vidgen, and M. Leveritt (2014). Sports dietitians Australia position statement: sports nutrition for the adolescent athlete. Int. J. Sport Nutr. Exerc. Metab. 24:570-584.
Ekeland, E., F. Heian, and K.B. Hagen (2005). Can exercise improve self esteem in children and young people? A systematic review of randomised controlled trials. Br. J. Sports Med. 39: 792-798.
Elhakeem, A., J. Heron, J.H. Tobias, and D.A. Lawlor (2020). Physical activity throughout adolescence and peak hip strength in young adults. J. Am. Med. Assoc. 3:e2013463.
Evans, M. Jr., H. Ndetan, M. Perko, R. Williams, and C. Walker (2012). Dietary supplement use by children and adolescents in the United States to enhance sport performance: results of the National Health Interview Survey. J. Primary Prev. 33:3-12.
Falk, B., O. Bar-Or, and J. MacDougall (1992). Thermoregulatory responses of pre-, mid-, and late-pubertal boys to exercise in dry heat. Med. Sci. Sports Exerc. 24:688-694.
Falk, B., and R. Dotan (2008). Children's thermoregulation during exercise in the heat: a revisit. Appl. Physiol. Nutr. Metab. 33:420-427.
Forbes, G.B. (1964). Growth of the lean body mass during childhood and adolescence. J. Pediatr. 64:822-827.
Grgic, O., E. Shevroja, B. Dhamo, A.G. Uitterlinden, E.B. Wolvius, F. Rivadeneira, and C. Medina-Gomez (2020). Skeletal maturation in relation to ethnic background in children of school age: The Generation R Study. Bone 132:115180.
Gymnastics Australia. (2019). Body Positive Guidelines.
Hirsch, K.R., A.E. Smith-Ryan, E.T. Trexler, and E.J. Roelofs (2016). Body composition and muscle characteristics of division I track and field athletes. J. Strength Cond. Res. 30:1231-1238.
Impey, S.G., M.A. Hearris, K.M. Hammond, J.D. Bartlett, J. Louis, G.L. Close, and J.P. Morton (2018). Fuel for the work required: A theoretical framework for carbohydrate periodization and the glycogen threshold hypothesis. Sports Med. 48:1031-1048.
Koehler, K., H. Braun, S. Achtzehn, U. Hildebrand, H.G. Predel, J. Mester, and W. Schanzer (2012). Iron status in elite young athletes: gender-dependent influences of diet and exercise. Eur. J. Appl. Physiol. 112:513-523.
Loucks, A.B. (2006). The response of luteinizing hormone pulsatility to 5 days of low energy availability disappears by 14 years of gynecological age. J. Clin. Endocrinol. Metab. 91:3158-3164.
Loureiro, L.L., S. Fonseca Jr., N.G. Castro, R.B. Dos Passos, C.P. Porto, and A.P. Pierucci (2015). Basal metabolic rate of adolescent modern pentathlon athletes: agreement between indirect calorimetry and predictive equations and the correlation with body parameters. PLoS One, 10:e0142859.
Lynch, B.A., T.K. Kaufman, T.I. Rajjo, K. Mohammed, S. Kumar, M.H. Murad, N.E. Gentile, G.A. Koepp, S.K. McCrady-Spitzer, and J.A. Levine (2019). Accuracy of accelerometers for measuring physical activity and levels of sedentary behavior in children: A systematic review. J. Prim. Care Community Health 10:2150132719874252.
Martinsen, M., S. Bratland-Sanda, A.K. Eriksson, and J. Sundgot-Borgen (2010). Dieting to win or to be thin? A study of dieting and disordered eating among adolescent elite athletes and nonathlete controls. Br. J. Sports Med. 44:70-76.
Mazzulla, M., K.A. Volterman, J.E. Packer, D.J. Wooding, J.C. Brooks, H. Kato, and D.R. Moore (2018). Whole-body net protein balance plateaus in response to increasing protein intakes during post-exercise recovery in adults and adolescents. Nutr. Metab. 15:62.
Mountjoy, M., J. Sundgot-Borgen, L. Burke, S. Carter, N. Constantini, C. Lebrun, N. Meyer, R. Sherman, K. Steffen, R. Budgett, and A. Ljungqvist (2014). The IOC consensus statement: beyond the Female Athlete Triad--Relative Energy Deficiency in Sport (RED-S). Br. J. Sports Med. 48:491-497.
Muia, E.N., H.H. Wright, V.O. Onywera, and E.N. Kuria (2016). Adolescent elite Kenyan runners are at risk for energy deficiency, menstrual dysfunction and disordered eating. J. Sports Sci. 34:598-606.
Orysiak, J., J. Mazur-Rozycka, J. Fitzgerald, M. Starczewski, J. Malczewska-Lenczowska, and K. Busko (2018). Vitamin D status and its relation to exercise performance and iron status in young ice hockey players. PLoS One 13:e0195284.
Peeling, P., T. Blee, C. Goodman, B. Dawson, G. Claydon, J Beilby, and A. Prins (2007). Effect of iron injections on aerobic-exercise performance of iron-depleted female athletes. Int. J. Sport Nutr. Exerc. Metab. 17:221-231.
Peeling, P., L.M. Castell, W. Derave, O. de Hon, and L.M. Burke (2019). Sports foods and dietary supplements for optimal function and performance enhancement in track-and-field athletes. Int. J. Sport Nutr. Exerc. Metab. 29:198-209.
Peeling, P., and V. Deakin (2021). Prevention, detection and treatment of iron depletion. In L. M. Burke, V. Deakin, & M. Minehan (Eds.), Clinical Sports Nutrition (6th ed.): McGraw Hill Education Australia.
Petrie, H., E. Stover, and C. Horswill (2004). Nutritional concerns for the child and adolescent competitor. Nutrition 20:620-631.
Randell, R.K., I. Rollo, T.J. Roberts, K.J. Dalrymple, A.E. Jeukendrup, and J.M. Carter (2017). Maximal fat oxidation rates in an athletic population. Med. Sci. Sports Exerc. 49:133-140.
Reale, R.J., T.J. Roberts, K.A. Lee, J.L. Bonsignore, and M.L. Anderson (2020). Metabolic rate in adolescent athletes: The development and validation of new equations, and comparison to previous models. Int. J. Sport Nutr. Exerc. Metab. 30:249-257.
Reguant-Closa, A., M.M. Harris, T,G, Lohman, and N.L. Meyer (2019). Validation of the athlete's plate nutrition educational tool: Phase I. Int. J. Sport Nutr. Exerc. Metab. 29:628-635.
Roche, D., T. Rowland, M. Garrard, S. Marwood, and V. Unnithan (2010). Skin microvascular reactivity in trained adolescents. Eur. J. Appl. Physiol. 108:1201-1208.
Rodenberg, R.E., and S. Gustafson (2007). Iron as an ergogenic aid: ironclad evidence? Curr. Sports Med. Rep. 6:258-264.
Rogers, M.A., R.N. Appaneal, D. Hughes, N. Vlahovich, G. Waddington, L.M. Burke, and M. Drew (2021). Prevalence of impaired physiological function consistent with Relative Energy Deficiency in Sport (RED-S): an Australian elite and pre-elite cohort. Br. J. Sports Med. 55:38-45.
Sardinha, L.B., and P.B. Judice (2017). Usefulness of motion sensors to estimate energy expenditure in children and adults: a narrative review of studies using DLW. Eur. J. Clin. Nutr. 71:1026.
SCAN (2010). Scientific Advisory Committee on Nutrition: Iron and Health. London: British Government.
Shoemaker, M.E., Z.M. Gillen, B.D. McKay, N.A. Bohannon, S.M. Gibson, K. Koehler, and J.T. Cramer (2019). Sex-specific relationships among iron status biomarkers, athletic performance, maturity, and dietary intakes in pre-adolescent and adolescent athletes. J. Int. Soc. Sports Nutr. 16:42.
Shoemaker, M.E., Z.M. Gillen, B.D. McKay, K. Koehler, and J.T. Cramer (2020). High prevalence of poor iron status among 8- to 16-year-old youth athletes: Interactions among biomarkers of iron, dietary intakes, and biological maturity. J. Am. Coll. Nutr. 39:155-162.
Torun, B. (2005). Energy requirements of children and adolescents. Public Health Nutr. 8:968-993.
WHO (1983). World Health Organization. Measuring change in nutritional status : guidelines for assessing the nutritional impact of supplementary feeding programmes for vulnerable groups. World Health Organization. https://apps.who.int/iris/handle/10665/38768
Witard, O., I. Garthe, and S. Phillips (2019). Dietary protein for training adaptation and body composition manipulation in track and field athletes. Int. J. Sport Nutr. Exerc. Metab. 29:165-174.








