VITAMIN D AND THE ATHLETE – AN OVERSIMPLIFIED, COMPLEX BIOLOGICAL PROBLEM
Published
March 2019
Author
Graeme L. Close, Richard Allison, Daniel Owens
Topics

KEY POINTS
• The vitamin D endocrine system is an increasingly more complex biological pathway than originally described in sport science literature.
• Many athletes have low total vitamin D (25[OH]D) concentrations.
• Supplementing with mega bolus doses of vitamin D may be detrimental to vitamin D metabolism.
• Measuring total 25[OH]D may no longer be the best marker of vitamin D status.
• Very low vitamin D concentrations can negatively affect athletic performance both directly and indirectly.
INTRODUCTION
Vitamin D is recognized as the ‘sunshine vitamin,’ due to the production of the majority of vitamin D3 (cholecalciferol) in the skin via sunlight (or more specifically, ultraviolet B radiation exposure). This absorbed radiation causes the cholesterol precursor, 7-dehydrocholesterol to form previtamin D3 (Figure 1). Spontaneous isomerization then alters the double bonds of the thermodynamically unstable previtamin D3 to
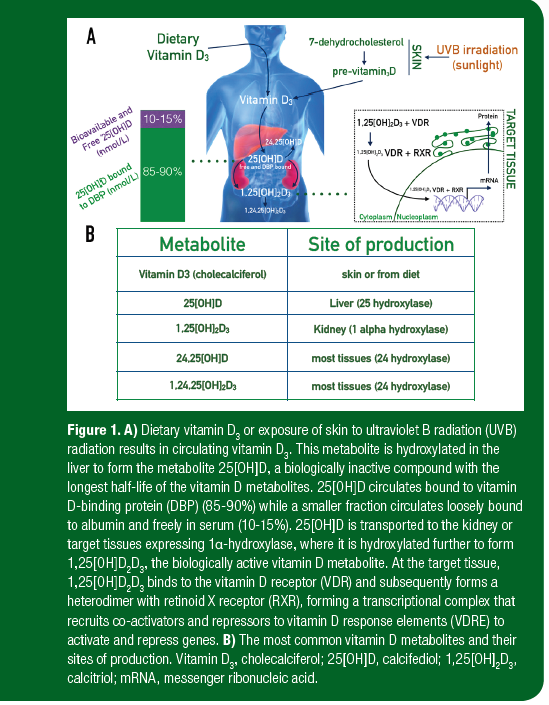
produce vitamin D3, also known as cholecalciferol (Holick, 2004). The significance of solar vitamin D production is clear when we consider that countries with low sunlight exposure for many months of the year have populations with the lowest vitamin D concentrations (Chen et al., 2007).
Vitamin D3 is mostly bound to the vitamin D-binding protein (DBP) in the circulation and is converted to 25-hydroxyvitamin D (25[OH]D, also known as calcifediol) in the liver under the control of the enzyme cytochrome P450 2R1 (CYP2R1) (Cheng et al., 2004; Shinkyo et al., 2004; Hamilton et al., 2010). Further hydroxylation of 25[OH]D to the active form of vitamin D, termed 1,25 dihydroxyvitamin D (1,25[OH]2D3, also known as calcitriol) occurs in the kidney (Figure 1). This bioactive metabolite is transported in the blood to the target tissues expressing the vitamin D receptor (VDR). It is this metabolite that exerts the effects of vitamin D in various tissues of the body, by binding to the VDR and subsequently regulating gene transcription.
Vitamin D status is typically categorized using serum 25[OH]D concentrations as severely deficient (< 12.5 nmol/L), deficient (12.5- < 30 nmol/L), insufficient (30-50 nmol/L) or sufficient (> 50 nmol/L) (The National Academies, 2011). However, the categorization of what constitutes 25[OH]D deficiency is widely debated (Owens et al., 2018). For now, it is therefore recommended to always present the absolute concentration of vitamin D as well as these arbitrary definitions.
HOW DOES VITAMIN D FUNCTION?
Research over the past two decades has established that the diverse biological actions of 1,25[OH]2D3 are driven through the control of gene expression, mediated by the VDR (McDonnell et al., 1987; Haussler et al., 1998) (Figure 1). Direct interaction between 1,25[OH]2D3 and the VDR leads to the interaction of the 1,25[OH]2D3-VDR complex with retinoid X receptor (RXR). This newly formed complex can then bind to vitamin D response elements located upstream of vitamin D target genes, and via the recruitment of co-activators and co-repressors, leads to the activation or repression of genes, respectively (Sutton & MacDonald, 2003). Historically, it was assumed that VDR-targeted gene expression was limited to the intestinal mucosa and bone. However, recent analyses indicate that vitamin D affects the expression of as much as 3% of the transcribed genome in target cells including those of the immune system, skin, the pancreas and skeletal muscle (Holick, 2007).
WHAT ARE THE CLAIMED BIOLOGICAL ROLES OF VITAMIN D RELEVANT TO ATHLETIC PERFORMANCE?
Athletic performance is determined by many genetic and environmental factors (MacArthur & North, 2005) and vitamin D has been proposed as both performance limiting and enhancing when in deficiency and abundance, respectively (Dueck et al., 1996; Cannell et al., 2009).
Bone Health
Recent studies have examined the association between vitamin D and bone and skeletal muscle function in athletes, two areas that may impact health and athletic performance. Vitamin D status is indicative of calcium absorption and bone mineralization (Berry et al., 2002) and there is considerable knowledge describing the relationship between 25[OH]D deficiency and bone health (Collins et al., 1998; Holick, 2006; Cashman et al., 2008; Breen et al., 2011; Gutierrez et al., 2011; Sadat- Ali et al., 2011; Wöfl et al., 2013). However, observations of vitamin D deficiency fail to universally affirm a proportionate susceptibility to bone loss, osteoporotic fractures or rickets (Hamson et al., 2003; Lowe et al., 2010), particularly in athletes, a population where stress fractures are frequently observed (Johnson et al., 1994). Interestingly, current research shows no association between 25[OH]D concentration and measures of bone health in an ethnically diverse athletic population, irrespective of exercise type (weight/non-weight bearing) (Allison et al., 2016) although others have demonstrated racial differences in manifestations for vitamin D and markers of bone health (Cauley et al., 2005; Hannan et al., 2008). What is clear is that bone cells are capable of producing 1,25[OH]2D3 from the 25[OH]D precursor and that this activity is likely to account for the skeletal effects of circulating 25[OH] D (Anderson & Atkins, 2008). To better understand the relationship between vitamin D and bone health in athletes, our group has turned to the examination of alternative vitamin D metabolites that may better reflect the biological activity of vitamin D (see ‘Measuring Vitamin D’ section).
Muscle Function and Remodeling
Elite athletes are known to be vitamin D deficient (Morton et al., 2012) and there is accumulating evidence exhibiting the role of vitamin D in skeletal muscle. Studies show conflicting results between vitamin D status and muscle strength and performance, and some demonstrate no relationships between 25[OH]D status and muscle strength (Dhesi, 2004; El-Hajj Fuleihan, 2005; Annweiler et al., 2009; Ceglia et al., 2011). However, Close et al. (2013) found an improvement in 10-m sprint times and vertical jump height following supplementation with 5,000 IU of D3/day. Sinha et al. (2013) also demonstrated that supplementing severely deficient athletes (< 15 nmol/L) with 20,000 IU D3 on alternate days significantly elevated phosphocreatine recovery half-time of the soleus muscle following activity, indicative of improved mitochondrial oxidative function. However, other studies have shown no improvement in muscle contractile properties in vitamin D-inadequate young active males following supplementation with 10,000 IU D3/day or placebo for three months (Owens et al., 2014). It would appear that in terms of muscle function, problems are only observed when athletes present with clinically low vitamin D concentrations (< 25 nmol/L) (Stockton et al., 2011). Therefore, it may be wise to focus on correcting deficiencies rather than trying to achieve supraphysiological concentrations.
Emerging evidence from integrative biology studies suggest that maintaining serum 25[OH]D concentrations > 50 nmol/L may be beneficial for skeletal muscle reparative processes and potentially for facilitating subsequent remodeling (Owens et al., 2015; 2017). Supplementation of vitamin D-inadequate men with 4,000 IU D3/day has been shown to improve muscle force recovery following a highvolume session of eccentric lower limb contractions (Owens et al., 2015). Moreover, skeletal myoblasts obtained from these individuals and ‘damaged’ in vitro showed improved muscle cell migration dynamics and improved myotube fusion/differentiation together with increased myotube hypertrophy. These data are the first to characterize a role for vitamin D in human skeletal muscle regeneration and suggest that maintaining serum 25[OH]D may be beneficial for enhancing reparative processes and potentially for facilitating subsequent hypertrophy (Owens et al., 2015).
Immune Health
An important factor that can limit athletic performance is poor immune health. Infections reduce playing time, selection availability and disrupt training programs. Vitamin D has long been known to modulate immune health in a variety of populations. A seminal study demonstrated that after adjusting for adiposity, lifestyle and socioeconomic factors, each 10 nmol/L increase in 25[OH]D was associated with a 7% reduction in infection risk (Berry et al., 2011). This could have important implications for athletes who, when overreaching or restricting nutrients such as carbohydrates, can compromise immune function (Gleeson & Walsh, 2012). Indeed, in an athletic cohort, supplemental vitamin D to increase serum 25[OH]D was sufficient to reduce infection risk during a 16- week period of winter training (He et al., 2013). A cause-effect relationship has also been determined for vitamin D and immune function, as both innate and acquired immune cells express the vitamin D receptor and vitamin D metabolizing enzymes and, importantly, are responsive to treatment with vitamin D (Hewison, 2012).
MEASURING VITAMIN D
Clinically, measuring serum 25[OH]D concentration provides the best estimate of vitamin D concentration (Holick, 1990; Iqbal, 1994) as both vitamin D3 (cholecalciferol) and 1,25[OH]2D3 have short half-lives (24 hours and 4-6 hours, respectively) and the circulating concentrations provide limited information about vitamin D status. In contrast, 25[OH] D has a long serum half-life (3 weeks) and the 25-hydroxylation step is unregulated, thus reflecting substrate availability. During vitamin D deficiency, parathyroid hormone increases and drives the renal 1-alphahydroxylase enzyme up, so that 1,25[OH]2D3 levels increase. Only in severe deficiency, when the substrate is depleted, does 1,25[OH]2D3 become low. Partially treated vitamin D deficiency also results in marked elevations of 1,25[OH]2D3 concentrations.
An Emerging Importance for Vitamin D-Binding Protein (DBP) and Bioavailable Vitamin D
In most clinical and athlete trials 25[OH]D concentration is measured as a marker of vitamin D status. However, there appears to be a ‘paradoxical relationship’ between ethnicity and vitamin D concentration that has largely been ignored, as black athletes generally have the lowest vitamin D concentrations but the greatest bone mineral density (BMD) and reduced risk of fracture (Cauley et al., 2005: Hannan et al., 2008). Vitamin D–binding protein (DBP) provides insight into why certain ethnic groups may have distinct 25[OH]D and BMD relationships (Powe et al., 2013). DBP is the primary vitamin D carrier, binding 85-90% of circulating 25[OH]D and 1,25[OH]2D3, the biologically active form of vitamin D, and the remaining unbound 25[OH]D is considered to be bioavailable. Bioavailable vitamin D is defined as 25[OH]D that is either free or bound to albumin. About 10-15% of total 25[OH]D is bound to albumin, in contrast to free 25[OH]D, which accounts for less than 1% of the total circulating vitamin D (Bikle et al., 1986). Since the affinity of albumin to 25[OH]D or 1,25[OH]2D3 is weaker than that of DBP, the loosely bound fraction and the free fraction consist of bioavailable 25[OH]D (Brown & Coyne, 2012).
DBP has many physiologically important functions including transporting vitamin D3 metabolites, binding/sequestering globular actin, and binding fatty acids. Genotyping has identified two common single-nucleotide polymorphisms (SNP) in the coding region of the DBP gene (rs4588 and rs7041) (Girgis et al., 2013). Combinations of these two SNPs produce three major polymorphic forms of DBP (Gc1F, Gc1S, and Gc2), which differ substantially in their binding affinity for 25[OH]D, circulating concentration, and variation between ethnic groups and are in turn linked to vitamin D binding protein function. Therefore, DBP may account for the observed racial differences in manifestations of vitamin D deficiency determined by 25[OH]D (Powe et al., 2013).
Studies have shown the association between serum 25[OH]D concentrations and numerous disease states and markers of athletic performance (Cannell et al., 2009; Holick, 2004). However, in an ethnically diverse athletic population, there was no relationship between serum 25[OH]D concentration and markers of bone health, regardless of sporting type (Allison et al., 2016). We have also recently shown that bioavailable vitamin D is a better predictor of bone mineral density (Allison et al., 2018). Systematic screening to determine 25[OH]D concentrations is expensive, and since it demonstrates a poor relationship to bone health in an ethnically diverse athletic population, it can be argued that in athletic communities, vitamin D testing should be reserved for the symptomatic athlete (i.e., musculoskeletal injury). This brings into question the validity of the commonly used laboratory test for 25[OH]D concentrations in assessing vitamin D deficiency and may provide insight as to why there is no currently universally accepted consensus for vitamin D concentrations. For researchers, the inclusion of DBP genotyping and measurement of free 25[OH]D would be beneficial for future trials hoping to analyze the relationship of 25[OH]D with parameters of athletic performance and athlete health. From an applied perspective, perhaps it is also time to develop tests to measure bioavailable vitamin D and, until this is available, blanket screening of athletes should be questioned. In terms of a target bioavailable vitamin D concentration, given that approximately 10% of total vitamin D is bioavailable, it could be argued that 5 nmol/L would be a suitable recommendation (based on a 50 nmol/L target concentration for total vitamin D) although data to support this suggestion is limited given that this is an emerging research topic.
SUPPLEMENTING WITH VITAMIN D: YOU CAN HAVE TOO MUCH OF A GOOD THING
The more evidence that research produces, the more complex is the task of correcting low vitamin D concentrations. It has often been suggested that 25[OH]D concentrations well above the U.S. Institute of Medicine guidelines are necessary for ‘optimal health’ (Zittermann, 2003; Heaney, 2013), but with little solid evidence to support this. As such, this idea has been followed in trials using large doses of vitamin D to achieve high 25[OH]D concentrations (> 100 nmol/L). Applied practitioners often translate this information into their practice, employing megadose supplementation, before such protocols have been fully scrutinized. Recent evidence refutes the suggestion that high concentrations of 25[OH]D are needed and that megadose supplements are efficacious. Work from our laboratory in conjunction with a professional squad of team sport athletes examined the metabolic responses of the vitamin D endocrine system following large doses of vitamin D, typically administered in elite team sport settings (Owens et al., 2017). Our results demonstrated that administration of a bolus dose containing 70,000 IU of vitamin D3 per week resulted in significant increases in vitamin D catabolism. Moreover, this increase in the inactive vitamin D metabolite outlived the decline in the active metabolites following withdrawal of supplementation, which could be potentially harmful and may explain the negative findings associated with megadose vitamin D supplementation (Sanders et al., 2010). A lower bolus of 35,000 IU/wk did not achieve such drastic changes in catabolism in the same trial. We believe that a moderate daily dose of vitamin D3 may therefore be most appropriate, if there is a need to supplement with vitamin D.
Moreover, as we have discussed in the current article, 25[OH]D may not even be the best marker to study the response to supplementation, since it may be the unbound fraction of 25[OH]D that best correlates with biological activity. Of importance, this fraction differs between races so it will be vital for new studies to include measurements of free/ bioavailable vitamin D to design the most efficacious supplementation strategies for vitamin D. The worry is that for now, we could be making an incorrect diagnosis of deficiency based on an unsuitable assay, then prescribing an untested but potentially deleterious dose of vitamin D.
PRACTICAL APPLICATIONS
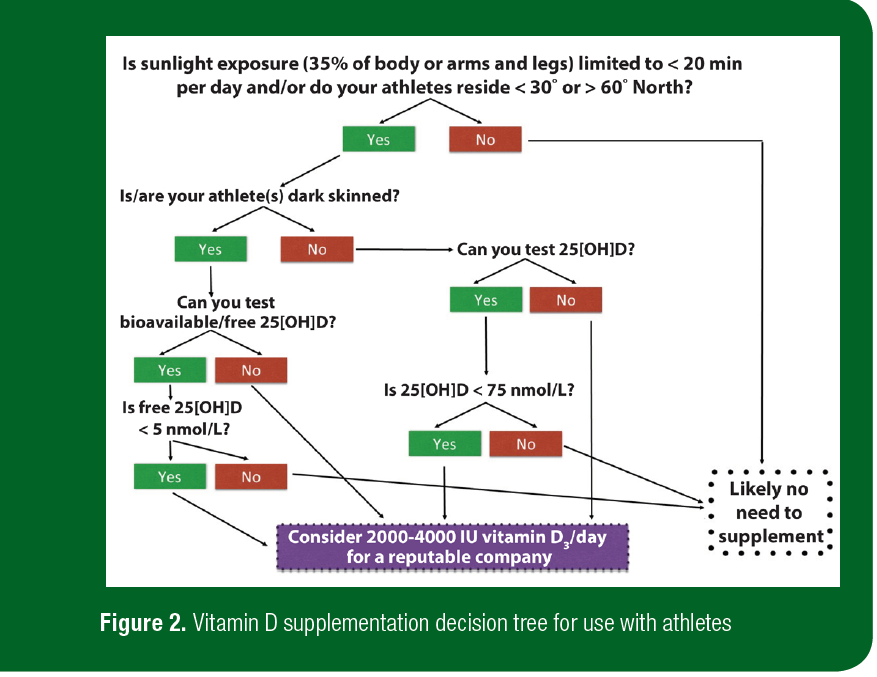
• The steps recommended to test and identify where vitamin D supplementation may be required can be observed in the decision flow chart observed in Figure 2.
• Based on current evidence, supplementing daily as opposed to weekly, with vitamin D3 in the range of 2,000-4,000 IU/day appears to pose no harmful effects and is within both the European and American safe upper limits for daily intake (The National Academies, 2011; European Food Safety Authority, 2012).
• Vitamin D3 should be used over the D2 form, as the latter has lower potency and its biological importance in humans is debated (Heaney et al., 2011). This strategy of D3 supplementation could be employed in climates where there is little sun exposure during winter months (October-March) or where climate, lifestyle and socioeconomic differences prevent sun exposure, such as in the Middle East, where vitamin D deficiency is prevalent (Hamilton et al., 2010).
• Where possible, seeking sensible sun exposure (10 a.m.-3 p.m., 15 min, 6 days/wk, 30-60ºN, ~30% body exposed, i.e., t-shirt and shorts) during the summer months may maintain adequate vitamin D concentrations without the need to supplement, even in climates such as Northern England (Rhodes, 2012).
SUMMARY
The past decade has witnessed considerable interest in vitamin D in athletic populations with vitamin D supplementation now becoming routine in many sporting teams and individual athletes. This interest has stemmed from numerous studies identifying ‘deficiencies’ in athletes. However, given the emerging research suggesting that our chosen method of assessment may not be appropriate to identify true deficiencies combined with evidence demonstrating potential deleterious effects of megadose supplementation, it may be time for a period of reflection and reassessment of current strategies. Vitamin D metabolism is a rapidly evolving field. We are moving inevitably closer to a more complete picture of this complex endocrine system, but much more is still to be learned. It is clear that clinically low vitamin D concentrations are detrimental to aspects of health that influence athletic performance, so it may be wise to attempt to avoid these deficiencies with low dose supplementation (~2,000 IU/day in the winter) and focus less on supplementation strategies to achieve ‘optimal’ concentrations. Research should move away from simply reporting that yet another group of athletes presents with ‘low’ 25(OH) D concentrations and rather focus upon free vitamin D and a better understanding of the true association between vitamin D and physiological function. A simple commercially available assay for free vitamin D needs to be developed but, until this is available, routine screening for 25[OH]D in athletes may be an expensive waste of time.
REFERENCES
Allison, R.J., A. Farooq, B. Hamilton, G.L. Close, and M.G. Wilson (2016). No association between vitamin D deficiency and markers of bone health in athletes. Med. Sci. Sports Exerc. 47:782- 788.
Allison, R. J., Farooq, A., Cherif, A., Hamilton, B., Close, G. L. and Wilson, M. G. (2018). Why don't serum vitamin D concentrations associate with BMD by DXA? A case of being 'bound' to the wrong assay? Implications for vitamin D screening. Br J Sports Med 52: 522-526.
Anderson, P.H., and G.J. Atkins (2008). The skeleton as an intracrine organ for vitamin D metabolism. Mol. Aspects Med. 29:397-406.
Annweiler, C., O. Beauchet, B. Fantino, G. Berrut, M. Bonnefoy, F.R. Herrmann, and A.M. Schott (2009). Is there an association between serum 25-hydroxyvitamin D concentration and muscle strength among older women? Results from baseline assessment of the EPIDOS study. J. Nutr. Health Aging 13:90-95.
Berry, J.L., M. Davies, and A.P. Mee (2002). Vitamin D metabolism, rickets, and osteomalacia. Semin. Musculoskelet. Radiol. 6:173-182.
Berry, D.J., K. Hesketh, C. Power, and E. Hypponen (2011). Vitamin D status has a linear association with seasonal infections and lung function in British adults. Br. J. Nutr. 106:1433-40.
Bikle, D.D., E. Gee, B. Halloran, M.A. Kowalski, E. Ryzen, and J.G. Haddad (1986). Assessment of the free fraction of 25-hydroxyvitamin D in serum and its regulation by albumin and the vitamin D-binding protein. J. Clin. Endocrinol. Metab. 63:954-959.
Breen, M.E., E.M. Laing, D.B. Hall, D.B. Hausman, R.G. Taylor, C.M. Isales, K.H. Ding, N.K. Pollock, M.W. Hamrick, C.A. Baile, and R.D. Lewis (2011). 25-hydroxyvitamin D, insulin-like growth factor-I, and bone mineral accrual during growth. Am. J. Clin. Endocrinol. Metab. 96:E89-E98.
Brown, A.J., and D.W. Coyne (2012). Bioavailable vitamin D in chronic kidney disease. Kidney Int. 82:5-7.
Cannell, J.J., B.W. Hollis, M.B. Sorenson, T.N. Taft, and J.J. Anderson (2009). Athletic performance and vitamin D. Med. Sci. Sports Exerc. 41:1102-1110.
Cashman, K.D., T.R. Hill, A.A. Cotter, C.A. Boreham, W. Dubitzky, L. Murray, J. Strain, A. Flynn, P.J. Robson, J.M. Wallace, and M. Kiely (2008). Low vitamin D status adversely affects bone health parameters in adolescents. Am. J. Clin. Nutr. 87:1039 1044.
Cauley, J.A., L.Y. Lui, K.E Ensrud, J.M. Zmuda, K.L Stone, M.C. Hochberg, and S.R. Cummings (2005). Bone mineral density and the risk of incident nonspinal fractures in black and white women. J. Am. Med. Assoc. 293:2102-2108.
Ceglia, L., G.R. Chiu, S.S. Harris, and A.B. Araujo (2011). Serum 25-hydroxyvitamin D concentration and physical function in adult men. Clin. Endocrinol. 74: 370-376.
Chen, T., F. Chimeh, Z. Lu, J. Mathieu, K.S. Person, A. Zhang, N. Kohn, S. Martinello, R. Berkowitz, and M.F. Holick (2007). Factors that influence the cutaneous synthesis and dietary sources of vitamin D. Arch. Biochem. Biophys. 460:213-217.
Cheng, J.B., M.A. Levine, N.H. Bell, D.J. Mangelsdorf, and D.W. Russell (2004) Genetic evidence that the human CYP2R1 enzyme is a key vitamin D 25-hydroxylase. Proc. Natl. Acad. Sci. USA 101: 7711-7715.
Collins, D., C. Jasani, I. Fogelman, and R. Swaminathan (1998). Vitamin D and bone mineral density. Osteoporos. Int. 8:110-114.
Close, G.L., J. Leckey, M, Patterson, W. Bradley, D.J. Owens, W.D. Fraser, and J.P. Morton (2013). The effects of vitamin D(3) supplementation on serum total 25[OH]D concentration and physical performance: a randomised dose-response study. Br. J. Sports Med. 47:692-696.
Dhesi, J.K. (2004). Vitamin D supplementation improves neuromuscular function in older people who fall. Age Ageing 33:589-595.
Dueck, C.A., K.S. Matt, M.M. Manore, and J.S. Skinner (1996). Treatment of athletic amenorrhea with a diet and training intervention program. Int. J. Sports Nutr. 6:24-40.
El-Hajj Fuleihan, G. (2005). Effect of vitamin D replacement on musculoskeletal parameters in school children: a randomized controlled trial. J. Clin. Endocrinol. Metab. 91:405-412.
European Food Safety Authority (2012). Scientific opinion on the tolerable upper intake level of vitamin D. EFSA J. 10:2813.
Girgis, C.M., R.J. Clifton-Bligh, M.W. Hamrick, M.F. Holick, and J.E. Gunton (2013). The roles of vitamin D in skeletal muscle: form, function, and metabolism. Endocrine Rev. 34:33–83.
Gleeson, M., and N.P. Walsh (2012). The BASES expert statement on exercise, immunity, and infection. J. Sports Sci. 30:321-324.
Gutierrez, O.M., W.R. Farwell, D. Kermah, and E.N. Taylor (2011). Racial differences in the relationship between vitamin D, bone mineral density, and parathyroid hormone in the National Health and Nutrition Examination Survey. Osteoporos, Int. 22:1745-1753.
Hamilton, B., J. Grantham, S. Racinais, and H. Chalabi (2010). Vitamin D deficiency is endemic in Middle Eastern sportsmen. Public Health Nutr. 13:1528-1534.
Hamson, C., L. Goh, P. Sheldon, and A. Samanta (2003). Comparative study of bone mineral density, calcium, and vitamin D status in the Gujarati and white populations of Leicester. Postgrad. Med. J. 79: 279-283.
Hannan, M.T., H.J. Litman, A.B. Araujo, C.E. McLennan, R.R. McLean, J.B. McKinlay, T.C. Chen, and M.F. Holick (2008). Serum 25-hydroxyvitamin D and bone mineral density in a racially and ethnically diverse group of men. J. Clin. Endocrinol. Metab. 93:40-46.
Haussler, M.R., G.K. Whitfield, C.A. Haussler, J.C. Hsieh, P.D. Thompson, S.H. Selznick, C.E. Dominguez, and P.W. Jurutka (1998). The nuclear vitamin D receptor: biological and molecular regulatory properties revealed. J. Bone Miner. Res. 13:325 349.
He, C.S., M. Handzlik, W.D. Fraser, A. Muhamad, H. Preston, A. Richardson, and M. Gleeson (2013). Influence of vitamin D status on respiratory infection incidence and immune function during 4 months of winter training in endurance sport athletes. Exerc. Immunol. Rev. 19:86-101.
Heaney, R.P., R.R. Recker, J. Grote, R.L. Horst, and L.A. Armas (2011). Vitamin D(3) is more potent than vitamin D(2) in humans. Am. J. Clin. Endocrinol. Metab. 96:E447 E452.
Heaney, R.P. (2013). Health is better at serum 25(OH)D above 30 ng/mL. J. Steroid. Biochem. Mol. Biol. 136:224-228.
Hewison, M. (2012). Vitamin D and immune function: an overview. Proc. Nutr. Soc. 71:50-61.
Holick, M.F. (1990). The use and interpretation of assays for Vitamin D and its metabolites. J. Nutr. 120 (Suppl. 11):1464-1469.
Holick, M.F. (2004).Vitamin D: importance in the prevention of cancers, type 1 diabetes, heart disease, and osteoporosis. Am. J. Clin. Nutr. 79:362-371.
Holick, M.F. (2006). Resurrection of vitamin D deficiency and rickets. J. Clin. Invest. 116:2062-2072.
Holick, M.F. (2007). Vitamin D deficiency. N. Engl. J. Med. 357:266-281.
Iqbal, S.J. (1994). Vitamin D metabolism and the clinical aspects of measuring metabolites. Ann. Clin. Biochem. 31:109-124.
Johnson, A.W., C.B. Weiss Jr., and D.L. Wheeler (1994). Stress fractures of the femoral shaft in athletes--more common than expected. A new clinical test. Am. J. Sports Med. 22:248-256.
Lowe, N.M., S.R. Mitra, P.C. Foster, I. Bhojani, and J.F. McCann (2010). Vitamin D status and markers of bone turnover in Caucasian and South Asian postmenopausal women living in the UK. Br. J. Nutr. 103:1706-1710.
MacArthur, D.G., and K.N. North (2005). Genes and human elite athletic performance. Human Gen. 116:331-339.
McDonnell, D.P., D.J. Mangelsdorf, J.W. Pike, M.R. Haussler, and B.W. O’Malley (1987). Molecular cloning of complementary DNA encoding the avian receptor for vitamin D. Science 235:1214-1217.
Morton, J.P., Z. Iqbal, B. Drust, D. Burgess, G.L. Close, and P.D. Brukner (2012). Seasonal variation in vitamin D status in professional soccer players of the English Premier League. Appl. Physiol. Nutr. Metab. 37:798-802.
Owens, D.J., D. Webber, S.G. Impey, J. Tang, T.F. Donovan, W.D. Fraser, J.P. Morton, and G.L. Close. (2014). Vitamin D supplementation does not improve human skeletal muscle contractile properties in insufficient young males. Eur. J. Appl. Physiol. 114:1309-1320.
Owens, D.J., A.P. Sharples, I. Polydorou, N. Alwan, T. Donovan, J. Tang, W.D. Fraser, R.G. Cooper, J.P. Morton, C. Stewart, and G.L. Close (2015). A systems-based investigation into vitamin D and skeletal muscle repair, regeneration, and hypertrophy. Am. J. Physiol. Endocrinol. Metab. 309:E1019-E1031.
Owens, D.J., J.C. Tang, W.J. Bradley, A.S. Sparks, W.D. Fraser, J.P. Morton, and G.L. Close (2017). Efficacy of high-dose vitamin D supplements for elite athletes. Med. Sci. Sports Exerc. 49:349-356.
Owens, D.J., R. Allison, and G.L. Close (2018). Vitamin D and the athlete: Current perspectives and new challenges. Sports Med. 48(Suppl 1):S3-S16.
Powe, C.E., M.K. Evans, J. Wenger, A.B. Zonderman, A.H. Berg, M. Nalls, H. Tamez, D. Zhang, I. Bhan, S.A. Karumanchi, N.R. Powe, and R. Thadhani (2013). Vitamin D binding protein and vitamin D status of black Americans and white Americans. N. Engl. J. Med. 369:1991-2000.
Rhodes, L.E. (2012) Maintaining vitamin D status in winter with ultraviolet B. Br. J. Dermatol. 166:238-239.
Sadat-Ali, M., A.H. Al Elq, H.A. Al-Turki, F.A. Al-Mulhim, and A.K. Al-Ali (2011). Influence of vitamin D levels on bone mineral density and osteoporosis. Br. J. Dermatol. 166:238-239.
Sanders, K.M., A.L. Stuart, E.J. Williamson, J.A. Simpson, M.A. Kotowicz, D. Young, and G.C. Nicholson (2010). Annual high-dose oral vitamin D and falls and fractures in older women: a randomized controlled trial. J. Am. Med. Assoc. 303:1815-1822.
Shinkyo, R., T. Sakaki, M. Kamakura, M. Ohta, and K. Inouye (2004). Metabolism of vitamin D by human microsomal CYP2R1. Biochem. Biophys. Res. Commun. 324 451 457.
Sinha, A., K.G. Hollingsworth, S. Ball, and T. Cheetham (2013). Improving the vitamin D status of vitamin D deficient adults is associated with improved mitochondrial oxidative function in skeletal muscle. Am. J. Clin. Endocrinol. Metab. 98:E509-E513.
Stockton, K.A., K. Mengersen, J.D. Paratz, D. Kandiah, and K.L. Bennell (2011). Effect of vitamin D supplementation on muscle strength: a systematic review and meta analysis. Osteoporos. Int. 23:859-871.
Sutton, A.L., and P.N. MacDonald (2003). Vitamin D: more than a “bone-a-fide” hormone. Mol. Endocrinol. 17: 777-791.
The National Academies, Dietary Reference Intakes for Calcium and Vitamin D (2011). Institute of Medicine. Vol. 1. 2011, Washington, DC: National Academic Press.
Wölfl, C., S. Englert, A.A. Moghaddam, G. Zimmermann, H. Schmidt-Gayk, B. Höner, A. Hogan, M. Lehnhardt, P.A. Grützner, and L. Kolios (2013). Time course of 25(OH)D3 vitamin D3 as well as PTH (parathyroid hormone) during fracture healing of patients with normal and low bone mineral density (BMD). BMC Musculoskel. Dis. 14:6.
Zittermann, A. (2003). Vitamin D in preventive medicine: are we ignoring the evidence? Br. J. Nutr. 89: 552-572.








