Nutritional Support for Injuries Requiring Reduced Activity

KEY POINTS
- The inflammatory response to an injury is an integral and important part of the healing process. Whereas excessive or prolonged inflammation may have negative consequences, the inflammatory response to most injuries is necessary and caution is warranted before attempts are made to counter it with nutritional interventions.
- Immobilization of a limb due to injury results in a sudden and dramatic loss of muscle mass, strength and function. Muscle loss with muscle inactivity results primarily from a global decrease in muscle protein synthesis and “anabolic resistance,” i.e., a diminished response to stimulation of muscle protein synthesis by anabolic or “muscle building” agents, including amino acids and insulin.
- Energy intake during a period of limb immobilization is often decreased from normal, but the decrease is unlikely to be as dramatic as may be initially considered. Careful determinations of energy expenditure and intake during recovery from injury are important aspects of nutritional management.
- A sudden and dramatic decrease in protein intake following a relatively severe injury – likely in conjunction with a voluntary decrease in energy intake – will lead to negative nitrogen balance resulting in impaired wound healing and/or increased muscle loss during periods of reduced activity. Maintenance, or even an increase, in the absolute amount of protein ingested during periods of reduced activity (g protein kg body mass-1 d-1) and energy intake are important aspects of nutritional support for injuries.
- There is a theoretical rationale and/or preliminary evidence for the efficacy of several nutraceuticals, including omega-3 fatty acids, leucine and creatine, to counter muscle loss during limb immobilization and/or reduced physical activity following injury. However, not only must any available data be considered preliminary, an understanding of the optimal dose, timing and potentially harmful consequences of using these nutraceuticals is in its infancy.
- There is also preliminary and indirect evidence for nutritional support for injuries other than those requiring limb immobilization, e.g., traumatic brain injuries (concussions) and muscle injuries. However, despite claims for the efficacy of many nutrients, in particular protein and free amino acid mixtures, most evidence is equivocal and it is not wise to make solid recommendations regarding these types of injury for any particular nutrient at this time.
INTRODUCTION
Every exerciser from elite-level athletes to those participating in physical activity for health and enjoyment can relate to the frustration of exercise-induced injury. Injuries range from very minor scrapes and bumps to more severe harm requiring prolonged inactivity and/or limb immobilization. Thus, there is a great deal of interest among sport and exercise practitioners to utilize interventions that can lessen the impact of injuries and increase the rate of healing and recovery. Among other interventions, nutritional strategies to support recovery from exercise-induced injuries have been considered. Unfortunately, despite the volume of material written about nutritional support for exercise-induced injury, most available information is based on speculation – even wishful thinking – and very few recommendations are backed by data from studies directly examining nutrition in injured humans (Tipton, 2015). This Sports Science Exchange article will discuss the evidence for support of injured athletes and exercisers through nutrition and attempt to make recommendations based on available information.
Recovery from exercise-induced injury may be considered to have two main stages, both of which may be influenced by nutrition choices. The first stage is the healing and recovery stage, characterized by an initial inflammatory response, wound healing and, almost certainly, reduced activity. More severe injuries may also result in limb immobilization and dramatically reduced activity levels. The second stage follows return to activity and rehabilitation. Often the lines between the two stages are somewhat blurry, but if limb immobilization is involved the transition from one stage to the other is usually rather clear. Unfortunately, it seems apparent that complete recovery, especially return to full activity and training, takes longer than the period of immobilization and/or reduced activity (Tipton, 2015). Clearly, appropriate nutritional support may be critical for a reduction in the negative impact of the injury per se, reduction in activity due to the injury and possibly immobilization of a limb, as well as to support increasing activity and rehabilitation. Nutritional recommendations for rehabilitation and return to full training are similar to those made for increased muscle growth (Phillips, 2014). Thus, this discussion primarily will focus on the first stage of recovery from injury, i.e., the time when wounds are healing and activity is reduced, possibly due to limb immobilization. Moreover, whereas most of the discussion will center on injuries leading to limb immobilization, an attempt to offer some information on other types of injuries also will be made.
INFLAMMATORY RESPONSE
An immediate inflammatory response is initiated following an exercise-induced injury. This response may last for a few hours up to many days depending on the severity of the injury (Calder, 2013). Whereas an appropriate inflammatory response is a critically important aspect of wound healing and thus recovery from injury, nutritional recommendations to lessen inflammation are commonly made. Recommendations for high intake of anti-inflammatory and antioxidant nutrients, such as omega-3 fatty acids (n-3FA), vitamins and phytonutrients are often touted (Calder, 2013). However, increasing intake of nutrients designed to curb inflammation following an injury should be done with caution. Given that the inflammatory response, per se, is critical for initiating optimal wound healing, dampening this response may be counterproductive. Moreover, important characteristics of supplementation designed to optimally reduce inflammation, such as the dose, frequency and duration of intake of nutrients, are unknown. Thus, unless there is evidence of excessive and overly prolonged inflammation – unlikely in otherwise healthy, injured athletes – caution is warranted regarding interventions aimed at eliminating inflammation.
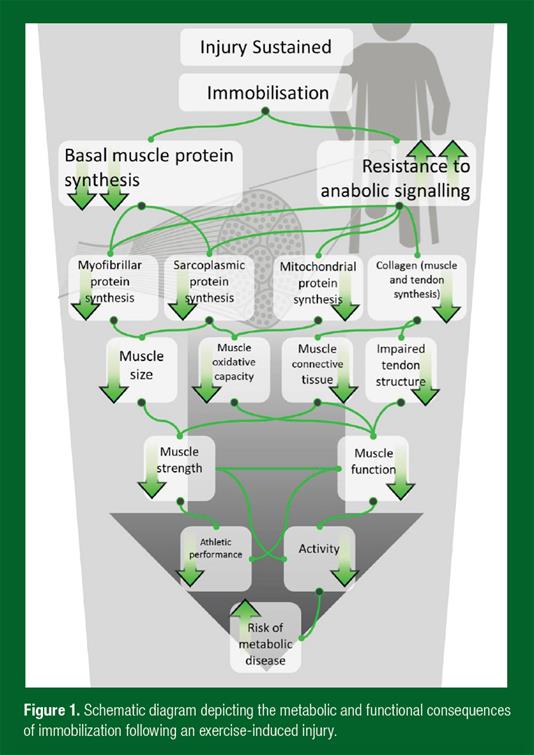
METABOLIC RESPONSES TO LIMB IMMOBILIZATION AND DECREASED ACTIVITY
One of the most onerous consequences of an exercise-induced injury is immobilization of a limb, forcing a drastic reduction in activity and training. Limb immobilization is harmful for tendon structure, muscle mass and tendon and muscle function of the injured limb and often results in a more general decline in activity levels. Measurable muscle loss has been reported in as little as 5 d of limb immobilization (Wall et al., 2014) and altered gene expression has been noted with only 48 h of muscle disuse (Reich et al., 2010). Thus, any injury requiring even limited immobilization and/or muscle disuse may have detrimental metabolic and functional consequences.
The metabolic mechanism for changes in muscle mass is net muscle protein balance (NBAL), i.e., the balance between the rate of muscle protein synthesis (MPS) and breakdown (MPB). Muscle is lost over any given period of time when intervals of negative NBAL are greater than intervals of positive NBAL. The preponderance of evidence suggests that changes in MPS are more influential on loss of muscle than changes in MPB. It is clear that during muscle disuse, the basal rate of MPS (resting and fasted) is decreased (Ferrando et al., 1996; Glover et al., 2008), but the influence of MPB on NBAL during muscle disuse is less clear. Measurement of MPB in humans is difficult and indirect measures are often necessary to attempt to assess changes in MPB that may contribute to muscle loss. After 14 d of strict bed rest, dynamic measurements of MPB using stable isotopic tracers show that MPB is decreased, albeit to a lesser extent than MPS (Ferrando et al., 1996). Thus, after 14 d the decrease in MPS is greater than that of MPB leading to negative NBAL and muscle loss. There is now preliminary and indirect evidence – based on mRNA expression of proteins involved in MPB pathways – that MPB may be increased during the first few days of immobility (Wall et al., 2014). These data suggest that a transient increase in MPB may contribute to muscle loss early after a limb is immobilized. However, increases in these indirect, static markers of MPB do not persist for longer periods, e.g., 14 d (Abadi et al., 2009; Glover et al., 2010). Moreover, there is convincing evidence that static, indirect markers of MPB do not represent the dynamic metabolism of muscle (Abadi et al., 2009; Glover et al., 2010; Phillips, 2014). Thus, solid conclusions regarding the importance of MPB for muscle loss during limb immobility are lacking. Nevertheless, it seems clear that decreased MPS is the major metabolic mechanism behind negative NBAL and muscle disuse atrophy.
Probably the most prominent metabolic contributor to muscle loss with disuse is the resistance of muscle to anabolic stimulation. The response of MPS to amino acids and exercise is reduced following a period of disuse and is termed “anabolic resistance.” It is clear that MPS does not respond as well to protein ingestion after disuse compared to when the muscle is active (Glover et al., 2008). Moreover, recent evidence suggests that simply reducing activity for 14 d leads to anabolic resistance in muscle (Breen et al., 2013). Thus, even if an injury does not result in full immobility of a limb, reduced activity as a result of the injury could lead to metabolic impairments and loss of muscle size and function. Nutritional interventions aimed to counter the reduction in basal MPS, as well as countering anabolic resistance of muscle, should be considered during periods of reduced activity and/or immobilization following an injury.
Muscle loss is not the only negative consequence of inactivity in muscle tissue. Muscle mitochondrial oxidative function and metabolic flexibility are also impaired with muscle disuse. Nearly all aspects of mitochondrial function are impacted (Abadi et al., 2009). Moreover, simply reducing activity of muscle for 14 d may lead to decreased insulin sensitivity of muscle (Breen et al., 2013). These adverse changes to muscle oxidative and metabolic function during immobilization are more evidence of the potential for the damaging impact of reduced muscle activity or immobilization following exercise-induced injuries.
NUTRITIONAL SUPPORT FOLLOWING INJURIES NECESSITATING IMMOBILIZATION
Many nutrients and nutritional strategies have been proposed to help improve recovery from exercise-induced injuries involving immobilization and/or reduced activity. A rationale for the use of many of these nutrients has been proposed, but little direct information is available (Tipton, 2015). A complete evaluation of all nutrients mentioned in this context is beyond the present scope and thus the focus will be on energy intake and protein/amino acids.
Energy Intake
Appropriate energy intake during immobilization is critical for optimal recovery and return to training. Energy expenditure almost certainly will decrease as a function of reduced training and activity, while immobilization of a limb involving ambulation may necessitate an even further decline. Nevertheless, the decrease in energy expenditure may not be as much as expected and careful consideration of energy intake should be made before recommendations are made to injured athletes.
There are several relatively subtle, and often underappreciated, factors that may influence energy expenditure and thus determine energy intake during limb immobilization and reduced activity. Injury, per se, increases energy expenditure. The magnitude of the increase, from 15 – 50%, and the duration of the increase depend entirely on the type and severity of the injury (Tipton, 2015). Furthermore, ambulation with crutches costs 2-3 times the energy of walking (Waters et al., 1987). Therefore, the total energy expenditure may not decrease as much as anticipated, particularly if the athlete does not voluntarily restrict movement during recovery.
An effort to attain energy balance during recovery from injury is critical. If restriction of energy intake is too severe, recovery almost certainly will be slowed due to reduced MPS, impaired wound healing (Tipton, 2015) and exacerbated muscle loss (Mettler et al., 2010). Thus, care should be taken to ensure that sufficient energy is consumed during recovery from an injury. On the other hand, a large positive energy balance also is undesirable for optimal healing and recovery. There is evidence that positive energy balance actually accelerates muscle loss during inactivity, most likely via activation of systemic inflammation (Biolo et al., 2008). Therefore, careful assessment of energy balance during both the period of inactivity and rehabilitation may be well worthwhile. Energy balance should be the aim during reduced inactivity and/or immobilization due to injury. Adjustment of energy intake to match expenditure as recovery progresses and the metabolic situation and activity patterns change is an important consideration.
Protein/Amino Acids
Protein is quite clearly the macronutrient most often associated with support for injuries involving immobilization and muscle loss (Tipton, 2015). Inadequate protein intake will impair wound healing and exacerbate muscle loss. Given that muscle loss results from decreased synthesis of myofibrillar proteins (Glover et al., 2008), and that the healing processes are heavily reliant on synthesis of collagen and other proteins, the importance of protein should be obvious. However, the optimal amount of protein in any given injury situation has yet to be determined.
The absolute amount of protein to be consumed on a daily basis is one important consideration for nutritional support of injuries. A reduction in protein intake, per se, may have a detrimental impact on muscle metabolism. This disruption may be particularly evident if habitual protein intake is relatively high (≥1.5 g protein kg body mass (BM)-1 d-1). A sudden decrease in protein intake results in negative nitrogen balance (Quevedo et al., 1994) and nitrogen loss is almost certainly from muscle during periods of negative energy balance (Pasiakos et al., 2010). It was demonstrated that athletes consuming relatively high protein intakes (~2.3g protein kg BM-1 d-1) had reduced muscle loss during periods of negative energy balance compared to athletes with lower protein intakes (~1.0 g protein kg BM-1 d-1) (Mettler et al., 2010). However, it is not clear whether the reduced muscle loss was due to protein intake above habitual (1.5 Þ 2.3 g protein kg BM-1 d-1). Rather, the loss of muscle in the lower protein group could have been due to the negative nitrogen balance when protein intake is lowered from habitual (1.5 Þ 1.0 g protein kg BM-1 d-1) (Quevedo et al., 1994). Taken together, it seems clear that protein intake should not decrease suddenly even in the face of reduced energy intake and that appropriate evaluation of habitual protein intake that helps inform recommendations for protein intake after injury should be made.
There are factors other than the total amount of protein to consider for optimal support of injury-induced muscle loss. MPS is maximized with the ingestion of ~20-25 g (Witard et al., 2014) or ~0.25-0.30 g/kg BM (Moore et al., 2014) in one dose of protein in both resting and contracted muscle . However, given the onset of anabolic resistance with immobility and reduced activity (Glover et al., 2008), it is likely that the amount of protein in each dose necessary to maximally stimulate MPS in immobilized muscle will be increased. Thus, another important consideration for nutritional support during recovery from injury should be careful consideration of the amount of protein in each meal.
The essential amino acid (EAA) content of the protein is likely the most important factor for optimal stimulation of MPS (Tipton et al., 1999). Thus, EAA supplementation has been suggested as a countermeasure for inactivity-induced muscle loss and has been shown to be effective for amelioration of muscle loss with inactivity (Paddon-Jones et al., 2004). Moreover, unlike many other proposed interventions, there has been direct assessment of decreased muscle loss with EAA supplementation following an injury (Dreyer et al., 2013). Thus, there is some evidence of efficacy of EAA supplementation during immobilization, but it is certainly not clear if EAA supplementation is more effective than consuming whole proteins containing the same amount of EAA. Given the cost (and taste) of EAA supplements, intact proteins may be a better choice for most injured athletes.
There is also evidence that the branched-chain amino acid, leucine, may be a particularly important EAA for amelioration of muscle loss with reduced activity, as it may be effective for overcoming muscle anabolic resistance (Katsanos et al., 2006). However, no study has directly investigated the response of muscle to leucine ingestion during a period of muscle disuse following an injury in humans. Moreover, there are potential negative effects, e.g., high blood ammonia, possible impairments in glucose control and brain neurotransmitters, with the use of high-dose leucine supplementation and so the optimal dose of leucine supplementation is unknown. Thus, caution is warranted prior to making recommendations for leucine supplementation during muscle disuse.
Evidence for efficacy of other nutrients during disuse atrophy
There is theoretical rationale for the efficacy for increased consumption of a variety of nutrients other than protein and amino acids during immobilization or reduced activity following injury. These nutrients include, but are not limited to, creatine, n-3FA and antioxidants. Creatine supplementation has been shown to decrease muscle atrophy in immobilized arms (Johnston et al., 2009), but not legs (Backx et al., 2017; Hespel et al., 2001). Given that creatine supplementation is widely used to enhance muscle gains during resistance exercise training (Hespel & Derave, 2007), it should be no surprise that creatine supplementation increased muscle gains following 10 weeks of rehabilitation after immobilization-induced muscle loss (Hespel et al., 2001). However, creatine did not enhance muscle regain with a shorter period of recovery from disuse-induced muscle atrophy (Backx et al., 2017). Thus, it seems that recommendations for creatine use by injured athletes may be situational, as the efficacy is clear during longer, but not shorter periods of muscle hypertrophy, while the impact on amelioration of muscle loss is uncertain.
Considerable support for use of n-3FA for nutritional support for injuries is available. In many cases, this attention is related to the anti-inflammatory and immunomodulatory properties of n-3FA (Calder, 2013). Furthermore, there is preliminary evidence suggesting that fish oil supplementation could play a role in the amelioration of muscle loss with disuse (You et al., 2010; Smith et al., 2011). At this time, there are no data showing that n-3FA supplementation effectively limits muscle loss in humans. Moreover, the appropriate dose for injured humans has not been established. Thus, wholesale recommendations for fish oil supplementation during immobilization must be considered premature and caution is warranted.
Oxidative damage is often a concern immediately following an injury. Oxidative damage is thought to be a contributing factor for muscle loss, primarily by increasing MPB (Magne et al., 2013). Thus, antioxidant compounds, including n-3FA, have been commonly recommended to improve healing and recovery (Demling, 2009; Magne et al., 2013). In high doses, there does seem to be some impact of antioxidant supplementation on muscle loss in rodents (Magne et al., 2013). However, equivalent doses would likely be problematic and potentially toxic if taken by humans (Magne et al., 2013). Lower doses that might be better tolerated tend not to be as effective. In one human study, vitamin C and E supplementation failed to influence recovery of muscle dysfunction following knee surgery (Barker et al., 2009). However, vitamin C status prior to supplementation was correlated with improvements in muscle function. Thus, taken together, these results suggest that sufficient antioxidant intake is important for optimal recovery, but supplementation on top of sufficiency is unnecessary if nutrient status is adequate.
NUTRITIONAL SUPPORT FOR OTHER INJURIES
Not all injuries require limb immobilization, and even if training is curtailed or reduced, muscle loss may be less and the metabolic consequences might not be as severe. Moreover, there is evidence that some injuries might have particular nutritional requirements. Unfortunately, to date very little is known about responses to nutrition in injured humans and/or any available data are equivocal. Nevertheless, a brief discussion of what little is known about nutrition to support a few selected types of injuries seems warranted. If for no other reason, mention of these areas is important to urge caution before wholeheartedly embracing a nutritional strategy touted by one of the many so-called experts.
Concussion (traumatic brain injury)
Traumatic brain injuries (TBI) in athletes are attracting an increasing amount of attention and scrutiny. In contact sports, such as rugby and American football, these injuries are increasingly common. However, there still are no approved therapies to treat TBI and the underlying processes and to enhance recovery from TBI (Barrett et al., 2014). Thus, it seems clear that a nutritional intervention that could ameliorate the consequences of TBI and improve cognitive and neuromuscular function would be valuable for active and retired athletes.
Antioxidant and anti-inflammatory agents are the focus of almost all nutrition-related research in relation to TBI. The research to date is based almost entirely on rodent models. Animal studies consistently demonstrate that both prophylactic and therapeutic use of n-3FA and curcumin increase cognitive function, as well as mechanistic indications of neural damage (Barrett et al., 2014). At this juncture, very little data have been published examining this question in humans. There have been a small number of case studies suggesting that high-dose n-3FA may improve acute outcomes after TBI. Recently, there have been promising results for the efficacy of docosahexanoic acid (DHA) supplementation, one of the n3-FA, for reducing markers of head trauma in American football players (Oliver et al., 2016). However, there is no consensus on the best biomarker for TBI, so the results from one study only should be viewed with caution. Interestingly, this study suggests that too much of a good thing may not be so good. Higher doses of DHA resulted in poorer results. So, not only is there uncertainty on the actual efficacy due to choice of biomarker, but the optimal dose needs to be determined. Thus, it is not clear if the efficacy of n-3FA for TBI in rodents can be applied appropriately to humans. Therefore, whereas preclinical and preliminary data on the impact of n-3FA for recovery from TBI are promising, solid recommendations to include n-3FA in a treatment regimen cannot be made, at least until the results of the ongoing clinical trials are reported.
Muscle Tissue Injuries
Common exercise-induced injuries include those with damaged muscle and other soft tissues. A common model used to examine muscle injuries is the eccentric (muscle lengthens while contracting) exercise model, which results in muscle soreness and loss of muscle function (Sousa et al., 2014). While nutrients have been suggested as countermeasures for muscle injuries, studies suggesting that protein and/or free amino acids may alleviate some indicators of muscle damage are most prominent. Other studies do not report an effect of protein or amino acids. The variable results are likely due to varying supplementation patterns, types of exercise and other design considerations (Sousa et al., 2014). Provision of antioxidants and anti-inflammatory agents to alleviate symptoms of muscle damage has been suggested as another popular strategy. However at best, as with protein, the literature can only be considered equivocal. Despite the equivocal nature of studies investigating the efficacy of protein, antioxidants and other nutrients, many recommendations are commonly made (Sousa et al., 2014). Clearly, given the disparity in the types of exercise, supplementation patterns and other methodological issues, very little insight into nutrition for muscle injuries can be gleaned from exercise-induced muscle damage studies. Hence, it is not possible to make solid recommendations regarding nutritional countermeasures to exercise-induced muscle damage and injuries. The reader is urged to exhibit caution before trying any particular nutritional method for athletes with muscle injuries.
WHAT TO AVOID
The main focus of the discussion above has been on what nutrients to consume. However, what to avoid also should be considered. As mentioned above, the most obvious nutritional consideration is to avoid nutrient deficiencies. On the other hand, excesses of nutrients also should be avoided.
Whereas many may consider it to be obvious, avoiding excessive alcohol intake is important following an injury. Alcohol ingestion impairs muscle protein synthesis (Vargas & Lang, 2008) and wound healing, likely by reducing the inflammatory response (Jung et al., 2011). Thus, at least in rats, alcohol intake increases muscle loss during immobilization (Vargas & Lang, 2008). Whereas it may be self-evident, it is worth emphasizing that limited alcohol ingestion during recovery is important. So, as tempting as it may be to indulge in alcohol to drown sorrows or diminish pain, only small amounts, if any, should be imbibed.
SUMMARY AND PRACTICAL APPLICATIONS
In summary, there is much still to be learned about the best nutritional strategy to enhance recovery from exercise-induced injuries. There are claims for the efficacy of many nutrients, yet direct evidence is sorely lacking. It is clear that a careful evaluation of each patient’s situation must be conducted before any nutritional recommendations are made to support recovery from exercise-induced injuries.
- The best recommendation would be to adopt a “first, do no harm” approach. The use and amount of each nutrient should be considered in the context of a risk/benefit ratio for each individual athlete with a particular injury situation. Even if the benefit is uncertain, it may be worth trying if no risks can be identified.
- As always, the basis of a nutritional strategy for an injured exerciser should be a well-balanced diet based on a diet of whole foods from nature (or foods made from ingredients from those foods) that are minimally processed.
- Deficiencies, particularly energy, protein and micronutrients, must be avoided. Energy balance is critical.
- Nutritional status and energy requirements should be assessed throughout recovery and nutrient intake adjusted accordingly.
- Higher protein intakes (~2-2.5 g protein kg BM-1 d-1) may be warranted. At the very least, the absolute amount of protein intake should be maintained even in the face of reduced energy intake.
- There is promising evidence for the efficacy of other nutrients, e.g., leucine, curcumin, n-3FA, in certain situations, but it must be considered preliminary. Caution is warranted before recommendations for wholesale use of these nutrients by injured athletes are made.
- At this point, solid recommendations for nutritional interventions as countermeasures for traumatic brain injuries and muscle injuries are premature. The same common sense nutritional recommendations apply for these injuries as those above.
REFERENCES
Abadi, A., E.I. Glover, R.J. Isfort, S. Raha, A. Safdar, N. Yasuda, J.J. Kaczor, S. Melov, A. Hubbard, X. Qu, S.M. Phillips, and M. Tarnopolsky (2009). Limb immobilization induces a coordinate down-regulation of mitochondrial and other metabolic pathways in men and women. PLoS One 4:e6518.
Backx EM, R. Hangelbroek, T. Snijders, M.L. Verscheijden, L.B. Verdijk, L.C. de Groot, and L.J. van Loon LJ (2017). Creatine loading does not preserve muscle mass or strength during leg immobilization in healthy, young males: a randomized controlled trial. Sports Med. E-pub ahead of print (PMID 28054322).
Barker, T., S.W. Leonard, J. Hansen, R.H. Trawick, R. Ingram, G. Burdett, K.M. Lebold, J.A. Walker, and M.G. Traber (2009). Vitamin E and C supplementation does not ameliorate muscle dysfunction after anterior cruciate ligament surgery. Free Rad. Biol. Med. 47:1611-1618.
Barrett, E.C., M.I. McBurney, and E.D. Ciappio (2014). Omega-3 fatty acid supplementation as a potential therapeutic aid for the recovery from mild traumatic brain injury/concussion. Adv. Nutr. 5:268-277.
Biolo, G., F. Agostini, B. Simunic, M. Sturma, L. Torelli, J.C. Preiser, G. Deby-Dupont, P. Magni, F. Strollo, P. di Prampero, G. Guarnieri, I.B. Mekjavic, R. Pisot, and M.V. Narici (2008). Positive energy balance is associated with accelerated muscle atrophy and increased erythrocyte glutathione turnover during 5 wk of bed rest. Am. J. Clin. Nutr. 88:950-958.
Breen, L., K.A. Stokes, T.A. Churchward-Venne, D.R. Moore, S.K. Baker, K. Smith, P.J. Atherton, and S.M. Phillips (2013). Two weeks of reduced activity decreases leg lean mass and induces "anabolic resistance" of myofibrillar protein synthesis in healthy elderly. J. Clin. Endocrinol. Metab. 98:2604-2612.
Calder, P.C (2013). n-3 fatty acids, inflammation and immunity: new mechanisms to explain old actions. Proc. Nutr. Soc. 72:326-336.
Demling, R.H (2009). Nutrition, anabolism, and the wound healing process: an overview. Eplasty 9:e9.
Dreyer, H.C., L.A. Strycker, H.A. Senesac, A.D. Hocker, K. Smolkowski, S.N. Shah, and B.A. Jewett (2013). Essential amino acid supplementation in patients following total knee arthroplasty. J. Clin. Invest. 123:4654-4666.
Ferrando, A.A., H.W. Lane, C. A. Stuart, J. Davis-Street, and R.R. Wolfe (1996). Prolonged bed rest decreases skeletal muscle and whole body protein synthesis. Am. J. Physiol. 270:E627-633.
Glover, E.I., S.M. Phillips, B.R. Oates, J.E. Tang, M.A. Tarnopolsky, A. Selby, K. Smith, and M.J. Rennie (2008). Immobilization induces anabolic resistance in human myofibrillar protein synthesis with low and high dose amino acid infusion. J. Physiol. 586:6049-6061.
Glover, E.I., N. Yasuda, M.A. Tarnopolsky, A. Abadi, and S.M. Phillips (2010). Little change in markers of protein breakdown and oxidative stress in humans in immobilization-induced skeletal muscle atrophy. Appl. Physiol. Nutr. Metab. 35:125-133.
Hespel, P., and W. Derave (2007). Ergogenic effects of creatine in sports and rehabilitation. Subcell. Biochem. 46:245-259.
Hespel, P., B. Op't Eijnde, M. Van Leemputte, B. Urso, P.L. Greenhaff, V. Labarque, S. Dymarkowski, P. Van Hecke, and E.A. Richter (2001). Oral creatine supplementation facilitates the rehabilitation of disuse atrophy and alters the expression of muscle myogenic factors in humans. J. Physiol. 536:625-633.
Johnston, A.P., D.G. Burke, L.G. MacNeil, and D.G. Candow (2009). Effect of creatine supplementation during cast-induced immobilization on the preservation of muscle mass, strength, and endurance. J. Strength Cond. Res. 23:116-120.
Jung, M.K., J.J. Callaci, K.L. Lauing, J.S. Otis, K.A. Radek, M.K. Jones, and E.J. Kovacs (2011). Alcohol exposure and mechanisms of tissue injury and repair. Alcohol Clin. Exp. Res. 35:392-399.
Katsanos, C.S., H. Kobayashi, M. Sheffield-Moore, A. Aarsland, and R.R. Wolfe (2006). A high proportion of leucine is required for optimal stimulation of the rate of muscle protein synthesis by essential amino acids in the elderly. Am. J. Physiol. 291:E381-387.
Magne, H., I. Savary-Auzeloux, D. Remond, and D. Dardevet (2013). Nutritional strategies to counteract muscle atrophy caused by disuse and to improve recovery. Nutr. Res. Rev. 26:149-165.
Mettler, S., N. Mitchelland K.D. Tipton (2010). Increased protein intake reduces lean body mass loss during weight loss in athletes. Med. Sci. Sports Exerc. 42:326-337.
Moore, D.R., T.A. Churchward-Venne, O. Witard, L. Breen, N.A. Burd, K.D. Tipton, and S.M. Phillips (2014). Protein ingestion to stimulate myofibrillar protein synthesis requires greater relative protein intakes in healthy older versus younger men. J. Gerontol. A Biol. Sci. Med. Sci. 70:57-62.
Oliver, J.M., M.T. Jones, K.M. Kirk, D.A. Gable, J.T. Repshas, T.A. Johnson, U. Andréasson, N. Norgren, Z. Blennow, and H. Zetterberg. (2016) Effect of docosahexaenoic acid on a biomarker of head trauma in American football. Med. Sci. Sport Exer. 48:974-982.
Paddon-Jones, D., M. Sheffield-Moore, R.J. Urban, A.P. Sanford, A. Aarsland, R.R. Wolfe, and A.A. Ferrando (2004). Essential amino acid and carbohydrate supplementation ameliorates muscle protein loss in humans during 28 days bedrest. J. Clin. Endocrinol. Metab. 89:4351-4358.
Pasiakos, S.M., L.M. Vislocky, J.W. Carbone, N. Altieri, K. Konopelski, H.C. Freake, J.M. Anderson, A.A. Ferrando, R.R. Wolfe, and N.R. Rodriguez (2010). Acute energy deprivation affects skeletal muscle protein synthesis and associated intracellular signaling proteins in physically active adults. J. Nutr. 140:745-751.
Phillips, S.M (2014). A brief review of critical processes in exercise-induced muscular hypertrophy. Sports Med. 44:S71-S77.
Quevedo, M.R., G.M. Price, D. Halliday, P.J. Pacy, and D.J. Millward (1994). Nitrogen homoeostasis in man: diurnal changes in nitrogen excretion, leucine oxidation and whole body leucine kinetics during a reduction from a high to a moderate protein intake. Clin. Sci. 86:185-193.
Reich, K.A., Y.W. Chen, P.D. Thompson, E.P. Hoffman, and P.M. Clarkson (2010). Forty-eight hours of unloading and 24 h of reloading lead to changes in global gene expression patterns related to ubiquitination and oxidative stress in humans. J. Appl. Physiol. 109:1404-1415.
Smith, G.I., P. Atherton, D.N. Reeds, B.S. Mohammed, D. Rankin, M.J. Rennie, and B. Mittendorfer (2011). Dietary omega-3 fatty acid supplementation increases the rate of muscle protein synthesis in older adults: a randomized controlled trial. Am. J. Clin. Nutr. 93:402-412.
Sousa, M., V.H. Teixeira, and J. Soares (2014). Dietary strategies to recover from exercise-induced muscle damage. Int. J. Food Sci. Nutr. 65:151-163.
Tipton, K.D. (2015). Nutritional support for exercise-induced injuries. Sports Med. 45:S93-S104.
Tipton, K.D., A.A. Ferrando, S.M. Phillips, D. Doyle Jr., and R.R. Wolfe (1999). Postexercise net protein synthesis in human muscle from orally administered amino acids. Am. J. Physiol. 276:E628-634.
Vargas. R., and C.H. Lang (2008). Alcohol accelerates loss of muscle and impairs recovery of muscle mass resulting from disuse atrophy. Alcoholism: Clin. Exp. Res. 32:128-137.
Wall, B.T., M.L. Dirks, T. Snijders, J.M. Senden, J. Dolmans, and L.J. van Loon (2014). Substantial skeletal muscle loss occurs during only 5 days of disuse. Acta Physiol. 210:600-611.
Waters, R.L., J. Campbell, and J. Perry (1987). Energy cost of three-point crutch ambulation in fracture patients. J. Orthop. Trauma 1:170-173.
Witard, O.C., S.R. Jackman, L. Breen, K. Smith, A. Selby, and K.D. Tipton (2014). Myofibrillar muscle protein synthesis rates subsequent to a meal in response to increasing doses of whey protein at rest and after resistance exercise. Am. J. Clin. Nutr. 99:86-95.
You, J.-S., M.-N. Park, W. Song, and Y.-S. Lee (2010). Dietary fish oil alleviates soleus atrophy during immobilization in association with Akt signaling to p70s6k and E3 ubiquitin ligases in rats. Appl. Physiol. Nutr. Metab. 35:310-318.








