MONITORING INTERNAL BODY TEMPERATURE
Published
May 2019
Author
Yasuki Sekiguchi; Luke N. Belval; Rebecca L. Stearns; Douglas J. Casa; Yuri Hosokawa

KEY POINTS
-
Accurately, conveniently, and quickly being able to monitor internal body temperature is an essential component of many research endeavors in
exercise science and is a critical diagnostic tool for determining exertional heat stroke and ruling out other potentially catastrophic medical conditions.
-
Rectal, gastrointestinal, and esophageal temperatures are the validated internal body temperature assessment methods that have application in research and/or sports medicine.
-
Oral, aural, tympanic, skin, axillary, and temporal temperature measurements should not be used to evaluate internal body temperature of an
exercising individual. -
The dual-heat-flux method (DHFM) and the zero-heat-flux (ZHF) method may prove useful to assess internal body temperature in real time during
exercise in the future. -
Prediction modeling of internal body temperature during exercise remains an approach which may increase performance and safety in the
physically active even though many technological and physiological problems must be overcome.
INTRODUCTION
Muscle Function and Remodeling
The assessment of internal body temperature is an essential component of exercise science research studies and is a key diagnostic measure in the field of sports medicine. The applications of monitoring body temperature can be far-reaching and can have important implications for athletes, soldiers, emergency service personnel and laborers. In the realm of exercise science research, it provides three critical components to the research process. First, it assures for the safety of the research subject during the study, an especially important measure when exercise is conducted in the heat or the exercise intensity is high, and especially when both conditions are present. Second, the measure allows for a critically dependent variable to be reported when evaluating research questions that may influence heat production. Examples in this area include studies investigating hydration status, beverage consumption, heat acclimatization, body cooling, environmental conditions, exercise intensity, equipment, clothing, fitness, etc. Third, temperature is often used to establish a steady state level of activity, as it can serve as an indicator of compensable heat stress, such that a particular research question can be evaluated.
In the sports medicine world, the valid assessment of internal body temperature provides information in at least four critical situations. First, and most importantly, is the assessment of internal body temperature during the entire process of care for exertional heat stroke (EHS). It is used first to make the diagnosis of EHS (Armstrong et al., 2007), then is used throughout cooling in cold water immersion to track the cooling process, and lastly is a critical measure to decide when to stop the cooling. It is safe to say that the immediate and valid measurement of internal body temperature has provided life-saving information for many exertional heat stroke survivors. Second, internal body temperature is routinely monitored during intense exercise in the heat as a preventative process, whether it be a special forces soldier who needs to simultaneously maintain intensity and safety, or an athlete returning from an EHS episode. The constant monitoring allows for the individual to stay within a safe degree of hyperthermia. Third, it is used regularly during the process of conducting heat tolerance tests. These are procedures where athletes/soldiers/laborers are provided an exercise heat challenge, and the measurement of the magnitude of hyperthermia is a factor in the assessment of the person’s exercise heat tolerance. Fourth, the internal body temperature can provide valuable information for an athlete with altered consciousness that does not have EHS. When the immediate assessment of internal body temperature does not reveal extreme hyperthermia then the athletic trainer or team physician can begin to consider other reasons for altered mental status, such as exertional hyponatremia, head injuries, hypoglycemia issues, cardiac issues (which should be assessed first before a body temperature measurement), exertional red blood cell sickling, or other possibilities
VALIDATED BODY TEMPERATURE ASSESSMENT METHODS
Previous studies suggest that esophageal, rectal and gastrointestinal thermometry are three measurement methods that may be used during exercise to monitor changes in internal body temperature (Bongers et al., 2018; Casa et al., 2007; Ganio et al., 2009; Hosokawa et al., 2016; 2017; Kolka et al., 1993). There are three vital components for body temperature assessment methods in exercising individuals: (1) ease of measurement (i.e., clinical external validity), (2) the measurement must be unaffected by the external environment (e.g., wind, solar radiation, sweat), and (3) the measurement accuracy and consistency are
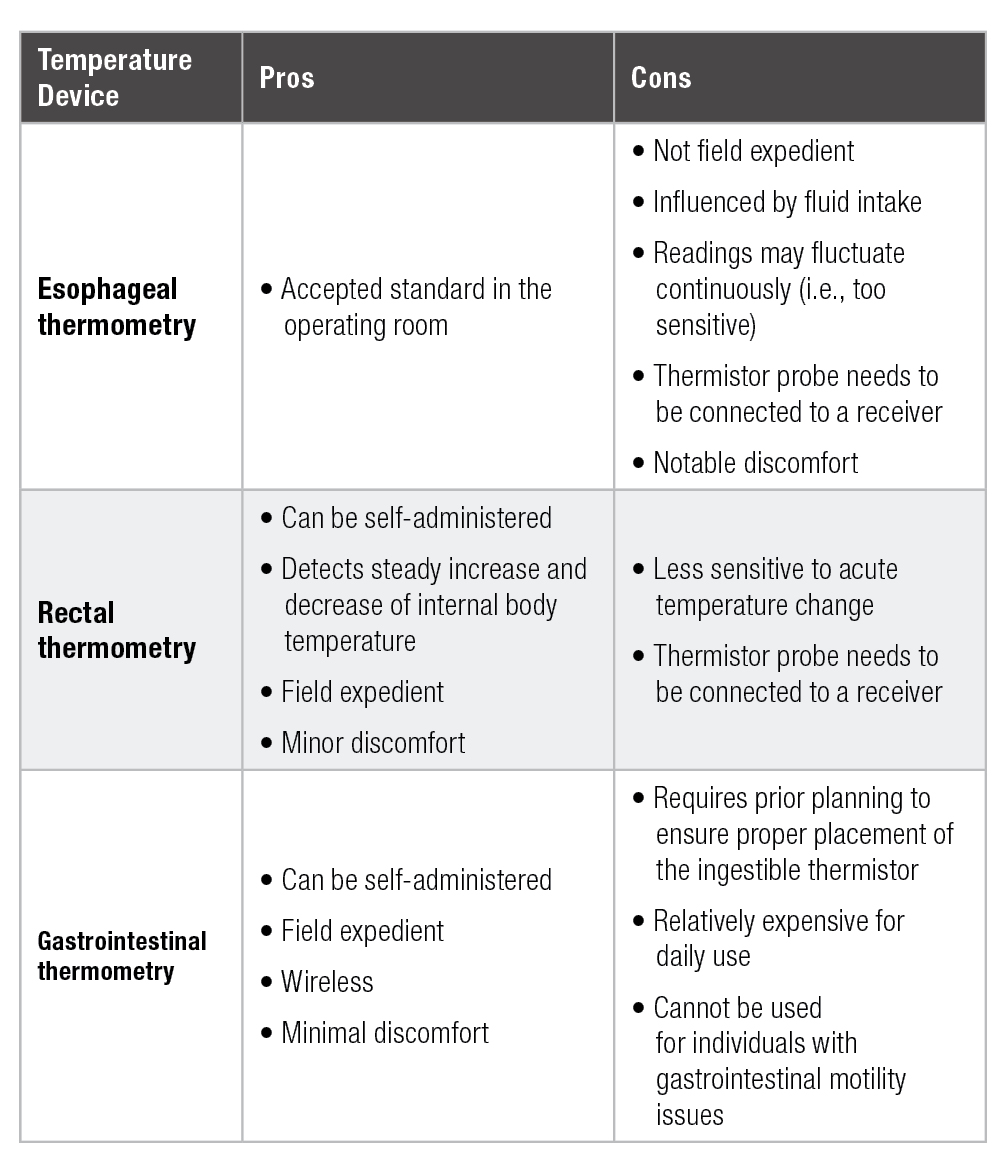
maintained throughout the exercise duration and post-exercise period (Casa et al., 2007; Ganio et al., 2009). Although these methods have been proven to be accurate, certain characteristics of each method warrants special consideration for field applications. The pros and cons of each method are summarized in Table 1.
Esophageal thermometry requires the insertion of a temperature probe through a nostril to the level of eighth and ninth thoracic vertebrae to measure the internal body temperature (Mekjavić & Rempel, 1990). Despite the common usage of esophageal thermometry in intraoperative procedures, the procedure for insertion requires specialized training and the application of the method within exercise is limited to laboratory-based studies (Hosokawa et al., 2017). Furthermore, the site o measurement may be too sensitive to internal body temperature changes such that clinical external validity may be limited (Gagnon et al., 2010; Hosokawa et al., 2017)
Rectal thermometry is the gold standard method for temperature assessment during and immediately post-exercise (Casa et al., 2007; Gagnon et al., 2010; Ganio et al., 2009). Compared to esophageal thermometry, rectal thermometry exhibits a steady increase and decrease of internal body temperature (Gagnon et al., 2010). The practical ease and validated accuracy of the technique also supports the use of rectal thermometry as the method for EHS diagnosis (Casa et al., 2015). While various forms of rectal thermometry exist, the most convenient form of the measurement contains a flexible probe (1–2 m) that allows for some room to prevent the probe from getting disconnected during physical movements. It should be noted that the insertion depth of the thermistor may influence the measurement, and it is recommended that one inserts the flexible probe to 15 cm (Miller et al., 2017). The application of continuous rectal temperature monitoring during exercise is mainly limited to distance running, where physical contact is minimal, although numerous published studies involving cycling in the heat have also employed rectal thermometry (Casa et al., 2007; Ganio et al., 2009; Hosokawa et al., 2017). Clothing attire and equipment in sports such as American football may also hinder clinicians from choosing rectal thermometry as a method for continuous internal body temperature assessment, as well as the need for the probe to be connected to a data logger (another practical inconvenience for field application).
Another method that is becoming more popular among sports scientists is gastrointestinal (GI) thermometry (Bongers et al., 2018; Hosokawa et al., 2016). GI thermometry uses a wireless, ingestible telemetric pill that contains a thermistor that transmits the internal body temperature readings to a receiver (Bongers et al., 2018; Casa et al., 2007; Ganio et al., 2009; Hosokawa et al., 2016). It has exhibited minimal mean bias (-0.1–0.2°C) when compared to rectal thermometry during exercise and the post-exercise period (Casa et al., 2007; Hosokawa et al., 2016). Improved devices are being introduced to the research community such that application in athletic settings may become more prevalent in the near future (Bongers et al., 2018). Importantly, in exchange for the convenience of wireless measurement, the use of an ingestible thermistor pill requires prior planning to ensure proper placement of the pill within the GI tract. The ingestion of the pill should be done at least 3 h prior to the exercise to minimize the chance of prematurely measuring gastric temperature and within 8 h to minimize the chance of pill passage (Casa et al., 2015; Ganio et al., 2009; Hosokawa et al., 2016). Furthermore, if not timed correctly, cold-fluid ingestion may also influence the temperature reading (Savoie et al., 2015), which can be further impacted by individual variations in gut motility. Therefore, close attention to the athlete’s ad libitum fluid ingestion behavior is warranted when interpreting the data.
INVALID BODY TEMPERATURE ASSESSMENT METHODS
While the importance of body temperature monitoring and the use of validated devices to assess body temperature is clear, maintaining knowledge of invalid body temperature assessment devices is also vital for appropriate research and clinical practice. It is important to clearly highlight that the validity of temperature measurement devices typically varies based on the population and setting that is being examined. Some devices may be accurate when used in a resting individual who has not been exercising. However, in an exercise scenario, the validity of temperature measurement devices is typically threatened by factors that may include air temperature or wind, physiological changes in skin functioning, and surrounding fluids (i.e., sweat, saliva, etc.). For the purposes of this article, the devices being discussed were evaluated within an exercise-induced state of hyperthermia, defined as body temperatures over 38.3°C (100.9°F).
When body temperature is elevated due to exercise, oral, aural (taken from the aural canal through the ear), tympanic (taken via a temperature sensor placed on the tympanic membrane of the ear), axillary and
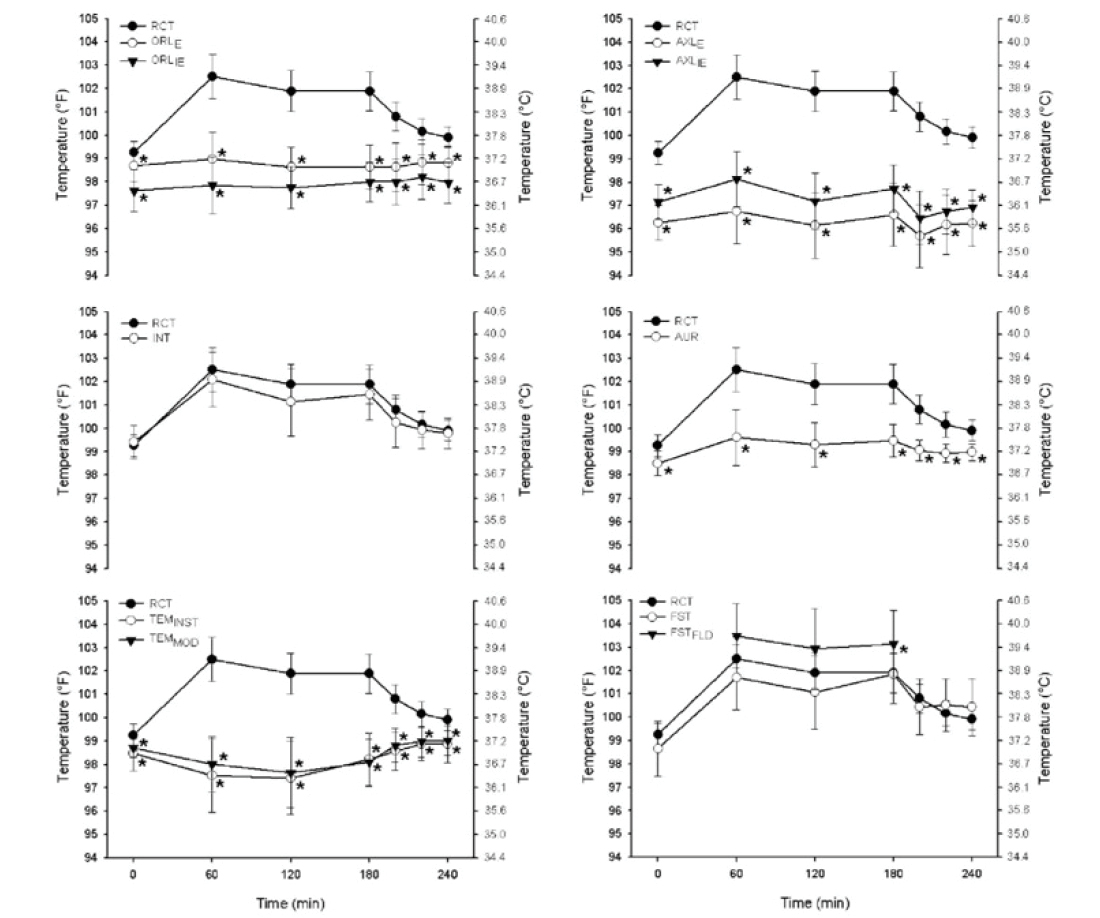
temporal (taken via a device that rolls across the forehead and temples) temperature measurement devices have all been found to be invalid forms of temperature assessment (Bagley et al., 2011; Casa et al., 2007; Ganio et al., 2009) (Figure 1). Variability in device validity exists when exercise-induced hyperthermia is low (37.0–38.5°C) (Fogt et al., 2017). Even with some research supporting the validity of these devices in low body temperature ranges, conflicting data continues to demonstrate that these devices provide unacceptable mean biases (considered above ± 0.27°C) with minimal exercise-induced body temperature elevations (rectal temperature < 38.5°C) (Bagley et al., 2011). This suggests that simply the introduction of exercise and elevated body temperature between 37.5–38.5°C, even in the presence of a cold environment, will provide invalid measures from these devices.
The validity of these devices with more severe body temperature elevations (rectal temperature > 38.5°C) is critical for proper diagnosis, treatment and survival for EHS patients. Unfortunately, many studies may not have reached these higher body temperatures and concluded that a device is valid in the absence of testing a full range of body temperatures. While these devices could be appropriate for use outside of an exercise situation, oral, aural, tympanic, axillary and temporal devices should not be used to evaluate the body temperature of an exercising individual.
WEARABLE TECHNOLOGIES TO MEASURE CORE TEMPERATURE
Continuous internal body temperature assessment provides time series data, which is the vital physiological information indicating the functioning of the body and the health condition. Consequently, the development of wearable technologies to measure internal body temperature has attracted many researchers. However, current methods do not allow real-time internal temperature measurements, especially when the internal body temperature is high. Some wearable technologies, which appear to have the potential to be used in the future following additional study, are discussed below.
The use of a thermometer based on the dual-heat-flux method (DHFM) is a relatively new method. The DHFM calculates internal body temperature based on the heat flow from the human body into a thermometer using at least four temperature sensors (Huang et al., 2016; 2017). Feng et al. (2017) reported that the difference of measured temperature between the DHFM and sublingual temperature was 0.13 ± 0.22°C at rest and 1.36 ± 0.44°C during exercise, while Huang et al. (2016) compared the DHFM to aural temperature. However, both the sublingual and aural temperature methods are not validated and are not the gold standard of internal body temperature assessment, and these studies have therefore not successfully demonstrated validation of the DHFM method. Huang et al. (2017) utilized the aural canal thermistor as the temperature reference when comparing to the DHFM. They demonstrated that the difference in measured temperature between the DHFM and the reference temperature was 0.07 ± 0.09°C during 55 min of rest and exercise (Huang et al., 2017). However, the internal body temperature did not exceed 38.0°C, so it remains questionable if the DHFM can be used to assess higher internal body temperatures. The zero-heat-flux (ZHF) method is another potential method to mea-sure internal body temperature. The ZHF sensor insulates the local skin surface, which is heated to deep body temperature to create a region of zero-heat flow from the body core to the skin (Teunissen et al., 2011). These authors demonstrated that ZHF tracked internal body temperature as measured by esophageal temperature with almost no time delay during exercise (the ZHF temperature - esophageal temperature = -0.05 ± 0.18°C) and recovery (the ZHF temperature - esophageal temperature = -0.01 ± 0.20°C) (Teunissen et al., 2011). However, internal body temperature did not exceed 38.5°C in this study as well; thus, the validity of higher internal body temperature measurement by ZHF remains to be tested. In addition to the DHFM and ZHF methods, Ota et al. (2017) demonstrated a 3D printed “earable” smart device to measure internal body temperature with an integrated bone conduction hearing aid. This device is designed to be worn on the ear to detect the temperature from the tympanic membrane based on an infrared sensor and the data is processed by an integrated module (Ota et al., 2017). However, the measured temperature with this device was again compared to tympanic temperature and skin temperature, which are not the gold standard of internal body temperature assessment. The DHFM and ZHF methods have the potential to measure internal body temperature in real time. However, they have not been validated against gold standard techniques and have not been tested when internal body temperature is higher than 38.5°C, which is easily reached during exercise, especially in the heat.
TECHNOLOGIES TO PREDICT CORE TEMPERATURE
With the advent of accessible wearable physiological sensors, the integration of signals with the purpose of predicting internal body temperature also has potential. Givoni and Goldman (1972) were among the first researchers who utilized prediction equations to overcome some of the obstacles associated with noninvasive temperature measurement. Since then, prediction models have become increasingly complex, integrating multiple sensors and mathematical equations, and in many cases leveraging the computational power of smartphones for potential use. Contemporary models can be separated into two main approaches – those that seek to explicitly model on the basis of the heat balance equation and those that model on integrated physiological responses.
Models that rely on the heat balance equation require instrumentation for both metabolic heat production and heat exchange with the environment. Common themes among published models include environmental temperature and humidity, skin temperature, and heart rate sensing requirements (Fiala et al., 2012; Kim & Lee, 2016; Niedermann et al., 2014; Richmond et al., 2015; Xu et al., 2013). The simplest models rely on only one or two measurements (Kim & Lee, 2016; Xu et al., 2013). Meanwhile, more complex models that utilize multiple skin temperature sites, heat flux measurements, heart rate, and metabolic measurements improve the predictive ability of these models but limit their potential application in field settings (Fiala et al., 2012; Niedermann et al., 2014; Richmond et al., 2015). Further detail on these models of thermophysiological stress can be found in Havenith and Fiala (2015).
More recently, models which consider a more integrative approach to thermal physiology have emerged. These models rely on the interactions between physiological systems to reduce the requirements for multiple sensors and assumptions. Principally, among these models are those which rely on sequential heart rate measurements to represent the strong interaction between the cardiovascular and thermoregulatory systems (Buller et al., 2013; 2018; Laxminarayan et al., 2018). While the sensor demands are greatly decreased with these models, greater computational complexity is gained to account for additional variability. Although these models are promising as they rely only on accurate heart rate measurements, like the previously mentioned models, they have yet to be fully validated.
While prediction models of internal body temperature have not achieved the necessary standard for the diagnosis and treatment of exertional heat illnesses (Moran & Mendal, 2002), other cases exist which may present opportunities for the prediction of internal body temperature. For example, adequately reliable prediction models could be used for the field assessment of acclimatization status, fulfilling a large gap in current technologies. In addition, future triage systems could leverage prediction models alongside other information to deliver appropriate emergent care for exertional heat illnesses. Despite the many technological and physiological problems that must be overcome, prediction modeling of internal body temperature during exercise remains an approach which may increase performance and safety in physically active individuals.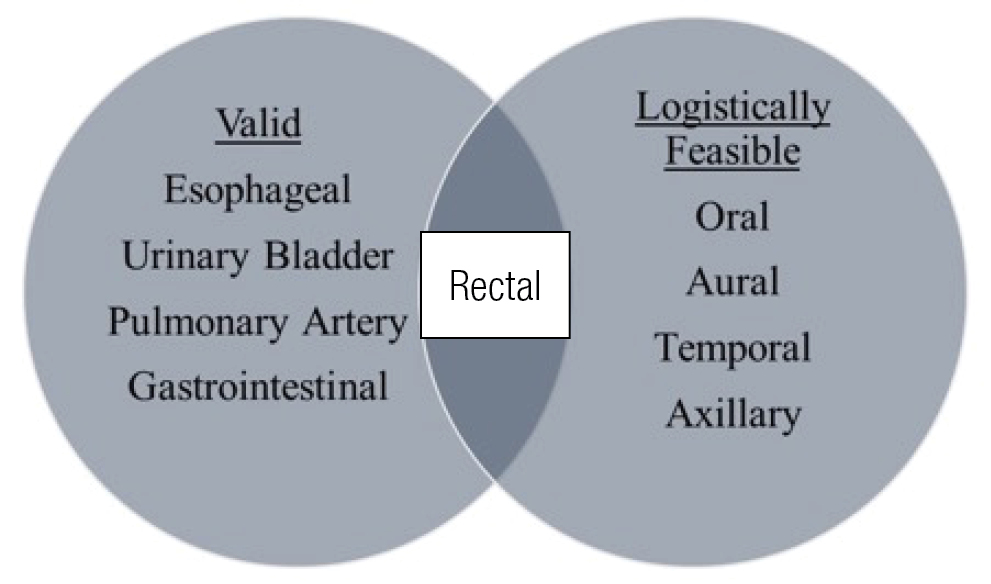
SUMMARY AND PRACTICAL APPLICATIONS
The issues of validity and feasibility are at the core when considering internal body temperature assessment (Figure 2). Many methods exist that are quite feasible, as can be seen in the right circle. Additionally, many methods exist that have the potential to be valid when measured correctly, as can be seen in the left circle. All internal devices are able to reflect internal body temperature. However, all the external devices are not accurate reflections of internal body temperature. The crux of the issue, at the point of the intersecting circles, is that only rectal temperature meets the strict standards of being valid and logistically feasible in both acute/emergency sports medicine and exercise science research scenarios. Rectal temperature, while certainly far from a perfect model for internal temperature measurement, allows for rapid recognition of EHS so that life-saving cooling can commence and continue until temperature is lowered to an acceptable level. Additionally, it offers a method to monitor temperature in laboratory settings where exercise science research is being conducted. It is clear that ingestible (GI) and esophageal temperature methods also have valuable applications in field and laboratory settings. However, they each have limitations when needed in other settings or when acute measures are needed in emergency settings (Savoie et al., 2015).
At this point we also await the emergence of new wearable technologies that accurately measure or predict internal body temperature. Currently, we must recommend that rectal temperature be the measurement tool of choice for assessment in EHS situations. In laboratory/research settings, multiple options exist, but depending on the circumstances are largely confined to rectal, esophageal, and GI (ingestible thermistors). The ingestible thermistors certainly have applications for preventative roles in sports medicine field settings, but rectal must be ready to be measured in emergency situations.
REFERENCES
Armstrong, L.E., D.J. Casa, M. Millard-Stafford, D.S. Moran, S.W. Pyne, and W.O. Roberts (2007). American College of Sports Medicine position stand. Exertional heat illness during training and competition. Med. Sci. Sports Exerc. 39:556–572.
Bagley, J.R., D.A. Judelson, B.A. Spiering, W.C. Beam, J.A. Bartolini, B.V. Washburn, K.R. Carney, C.X. Muñoz, S.W. Yeargin, and D.J. Casa (2011). Validity of field expedient devices to assess core temperature during exercise in the cold. Aviat. Space Environ. Med. 82:1098–1103.
Bongers, C.C.W.G., H.A.M. Daanen, C.P. Bogerd, M.T.E. Hopman, and T.M.H. Eijsvogels (2018). Validity, reliability, and inertia of four different temperature capsule systems. Med. Sci. Sports Exerc. 50:169–175.
Buller, M.J., W.J. Tharion, S.N. Cheuvront, S.J. Montain, R.W. Kenefick, J. Castellani, W.A. Latzka, W.S. Roberts, M. Richter, O.C. Jenkins, and R.W. Hoyt (2013). Estimation of human core temperature from sequential heart rate observations. Physiol. Meas. 34:781–798.
Buller, M.J., A.P. Welles, and K.E. Friedl (2018). Wearable physiological monitoring for human thermal-work strain optimization. J. Appl. Physiol. 124:432–441.
Casa, D.J., S.M. Becker, M.S. Ganio, C.M. Brown, S.W. Yeargin, M.W. Roti, J. Siegler, J.A. Blowers, N.R. Glaviano, R.A. Huggins, L.E. Armstrong, and C.M. Maresh (2007). Validity of devices that assess body temperature during outdoor exercise in the heat. J. Athl. Train. 42:333– 342.
Casa, D.J., J.K. DeMartini, M.F. Bergeron, D. Csillan, E.R. Eichner, R.M. Lopez, M.S. Ferrara, K.C. Miller, F. O’Connor, M.N. Sawka, and S.W. Yeargin (2015). National Athletic Trainers’ Association Position Statement: Exertional heat illnesses. J. Athl. Train. 50:986–1000.
Feng, J., C. Zhou, C. He, Y. Li, and X. Ye (2017). Development of an improved wearable device for core body temperature monitoring based on the dual heat flux principle. Physiol. Meas. 38:652–668.
Fiala, D., G. Havenith, P. Bröde, B. Kampmann, and G. Jendritzky (2012). UTCI-Fiala multi-node model of human heat transfer and temperature regulation. Int. J. Biometeor. 56:429–441.
Fogt, D.L., A.L. Henning, A.S. Venable, and B.K. McFarlin (2017). Non-invasive measures of core temperature versus ingestible thermistor during exercise in the heat. Int. J. Exerc. Sci. 10:225–233.
Gagnon, D., B.B. Lemire, O. Jay, and G.P. Kenny (2010). Aural canal, esophageal, and rectal temperatures during exertional heat stress and the subsequent recovery period. J. Athl. Train. 45:157–163.
Ganio, M.S., C.M. Brown, D.J. Casa, S.M. Becker, S.W. Yeargin, B.P. McDermott, L.M. Boots, P.W. Boyd, L.E. Armstrong, and C.M. Maresh (2009). Validity and reliability of devices that assess body temperature during indoor exercise in the heat. J. Athl. Train. 44:124–135.
Givoni, B., and R.F. Goldman (1972). Predicting rectal temperature response to work, environment, and clothing. J. Appl. Physiol. 32:812–822.
Havenith, G., and D. Fiala (2015). Thermal indices and thermophysiological modeling for heat stress. Compr. Physiol. 6:255–302.
Hosokawa, Y., W.M. Adams, R.L. Stearns, and D.J. Casa (2016). Comparison of gastrointestinal and rectal temperatures during recovery after a warm-weather road race. J. Athl. Train. 51:382–388.
Hosokawa, Y., W.M. Adams, and D.J. Casa (2017). Comparison of esophageal, rectal, and gastrointestinal temperatures during passive rest after exercise in the heat: the influence of hydration. J. Sport Rehab. 26:1–10.
Huang, M., T. Tamura, T. Yoshimura, T. Tsuchikawa, and S. Kanaya (2016). Wearable deep body thermometers and their uses in continuous monitoring for daily healthcare. Conf. Proc. IEEE Eng. Med. Biol. Soc. 177-180.
Huang, M., T. Tamura, Z. Tang, W. Chen, and S. Kanaya (2017). A wearable thermometry for core body temperature measurement and its experimental verification. IEEE J Biomed Health Inform. 21:708-714.
Kim, S., and J.Y. Lee (2016). Skin sites to predict deep-body temperature while wearing firefighters’ personal protective equipment during periodical changes in air temperature. Ergonomics 59:496–503.
Kolka, M.A., M.D. Quigley, L.A. Blanchard, D.A. Toyota, and L.A. Stephenson (1993). Validation of a temperature telemetry system during moderate and strenuous exercise. J. Therm. Biol. 18:203–210.
Laxminarayan, S., V. Rakesh, T. Oyama, J.B. Kazman, R. Yanovich, I. Ketko, Y. Epstein, S. Morrison, and J. Reifman (2018). Individualized estimation of human core body temperature using noninvasive measurements. J. Appl. Physiol. 124:1387–1402.
Mekjavić, I.B., and M.E. Rempel (1990). Determination of esophageal probe insertion length based on standing and sitting height. J. Appl. Physiol. 69:376–379.
Miller, K.C., L.E. Hughes, B.C. Long, W.M. Adams, and D.J. Casa (2017). Validity of core temperature measurements at 3 rectal depths during rest, exercise, cold-water immersion, and recovery. J. Athl. Train. 52:332–338.
Moran, D.S., and L. Mendal (2002). Core temperature measurement: methods and current insights. Sports Med. 32:879–885.
Niedermann, R., E. Wyss, S. Annaheim, A. Psikuta, S. Davey, and R.M. Rossi (2014). Prediction of human core body temperature using non-invasive measurement methods. Int. J. Biometeor. 58:7–15.
Ota, H., M. Chao, Y. Gao, E. Wu, L. Tai, K. Chen, Y. Matsuoka, K. Iwai, H. Fahad, W. Gao, H. Nyein, L. Lin, and A. Javey (2017). 3D printed “earable” smart devices for real-time detection of core body temperature. ACS Sensors 2:990–997.
Richmond, V.L., S. Davey, K. Griggs, and G. Havenith (2015). Prediction of core body temperature from multiple variables. Ann. Occup. Hyg. 59:1168–1178.
Savoie, F.A., T. Dion, A. Asselin, C. Gariepy, P.M. Boucher, F. Berrigan, and E.D. Goulet (2015). Intestinal temperature does not reflect rectal temperature during prolonged, intense running with cold fluid ingestion. Physiol. Meas. 36:259–272.
Teunissen, L.P., J. Klewer, A. de Haan, J.J. de Koning, and H.A. Daanen (2011). Non-invasive continuous core temperature measurement by zero heat flux. Physiol. Meas. 32:559–570.
Xu, X., A.J. Karis, M.J. Buller, and W.R. Santee (2013). Relationship between core temperature, skin temperature, and heat flux during exercise in heat. Eur. J. Appl. Physiol. 113:2381– 2389.








