Physiological Adaptations to Low-Volume High-Intensity Interval Training

KEY POINTS
- High-intensity interval training (HIIT) is generally characterized by repeated sessions of brief, intermittent exercise, typically at intensities that elicit ≥85% of peak oxygen uptake (VO2peak), and interspersed by periods of rest or low-intensity exercise for recovery.
- While long appreciated by endurance athletes as an integral component of training programs designed to maximize performance, short-term studies lasting up to several weeks in healthy persons of average fitness have established that HIIT per se is a potent stimulus to induce physiological adaptations that resemble changes typically associated with traditional endurance training, despite a lower total exercise volume and reduced training time commitment.
- As little as six sessions of HIIT over 2 wk, using a protocol that entails only 2-3 min of all-out exercise within a training session that lasts ~20 min (i.e., repeated Wingate Tests), can increase skeletal muscle oxidative capacity, reduce non-oxidative energy provision during submaximal exercise and markedly improve performance during tasks that rely mainly on aerobic energy metabolism.
- While all-out HIIT protocols are very effective, other low-volume HIIT models that consist of relatively intense, but submaximal, constant-load efforts (e.g., 10 x 60 s at a fixed work intensity that elicits ~90% of maximal heart rate, interspersed by 60 s of recovery) have been shown to induce rapid physiological and performance adaptations similar to Wingate-based training.
- The majority of low-volume HIIT studies conducted to date have utilized relatively short intervention periods (i.e., lasting up to several weeks) and future work involving long-term (i.e., months to years) interventions is needed to advance our mechanistic understanding of how manipulating the exercise stimulus translates into physiological remodeling, as well as identifying from a practical perspective the minimum “dose” of HIIT to maximize adaptation, given that lack of time remains the most commonly cited barrier to lack of regular exercise participation.
INTRODUCTION
Regular endurance training improves performance during tasks that rely mainly on aerobic energy metabolism, in large part by increasing the body’s ability to transport and utilize oxygen, and enhancing the capacity for the oxidative metabolism of substrates by working skeletal muscle (Saltin & Gollnick, 1983). While less widely appreciated, high-intensity interval training (HIIT) is a potent stimulus to induce physiological adaptations that resemble, and indeed may be superior to, changes typically associated with traditional endurance training (Kubukeli et al., 2002; Ross & Leveritt, 2001). Indeed, highly-trained endurance athletes have long incorporated HIIT as an integral component of training programs designed to maximize performance (Laursen & Jenkins, 2002). Recently, short-term studies lasting up to several weeks in healthy persons of average fitness have established that HIIT per se is a potent stimulus to induce physiological adaptations that resemble changes typically associated with traditional endurance training, despite a lower total exercise volume and reduced training time commitment (Burgomaster et al., 2005; Gibala et al., 2006; Little et al., 2010). This brief review highlights recent work that sheds new light on the potency of low-volume HIIT to induce rapid physiological remodeling and enhance the capacity for performance during tasks that rely mainly on aerobic energy metabolism. For a more comprehensive analysis, as well as the potential application of HIIT to different populations, the reader is referred to other recent reviews by the present author (Gibala et al., 2012, 2014) and others, including work that has specifically focused on those at risk for, or afflicted by, cardiometabolic disorders (Kessler et al., 2012; Weston et al., 2014). With regard to practical applications and training prescription, two other recent reviews (Buchheit & Laursen, 2013ab) consider in detail various aspects of HIIT programming, with a particular focus on athletic performance.
RESEARCH REVIEW – WHAT IS HIIT?
High-intensity interval training (HIIT) is generally characterized by repeated sessions of brief, intermittent exercise, typically at intensities that elicit ≥85% of peak oxygen uptake (VO2peak), and interspersed by periods of rest or low-intensity exercise for recovery.
A wide range of terms have been used to describe various interval training protocols, leading to many different acronyms and a general lack of standardization in the literature. A classification scheme was recently proposed in which the term “HIIT” is used to describe protocols in which the training stimulus is “near maximal” or the target intensity is between 80-100% of maximal heart rate, and “sprint interval training” (SIT) be used for protocols that involve “all -out” or “supramaximal” efforts, in which target intensities correspond to workloads greater than what is required to elicit maximal oxygen uptake or (VO2peak) (Weston et al., 2014).
The term "HIIT" will be used exclusively in this review for simplicity, but the interested reader is referred to Weston et al. (2014) for further consideration. Similarly, there is no universal definition of what constitutes “low-volume” interval training, but we have generally considered this to be protocols in which the total amount of intense exercise performed during a training session is ≤10 min, i.e., the summed total duration of the hard efforts, excluding the recovery periods and any warm-up or cool-down (Gibala et al., 2014). With respect to studies that have examined physiological adaptations to low-volume HIIT, one of the most common protocols employed is the Wingate Test, which involves 30 s of maximal cycling on a specialized ergometer, typically using a braking force or resistance that is equivalent to 7.5% of body mass. The task is extremely demanding, and during a single effort subjects typically generate mean power output values that correspond to ~250-300% of what can be achieved during a standard incremental test to determine (VO2peak). A single training session lasts ~20-25 min including brief warm-up and cool-down, with subjects typically performing 4-6 Wingate Tests separated by a few minutes of recovery (Burgomaster et al., 2005; Gibala et al., 2006).
Another common intervention employed in low-volume HIIT studies is repeated constant-load efforts performed at a high relative (but not all-out) work intensity; for example, ~10 60-s cycling efforts at 100% of the peak power output elicited during a ramp VO2peak test, or an intensity that elicits ~90% of maximal heart rate, interspersed with a similar amount of recovery between efforts (Little et al., 2010). An overview of some common protocols employed in interval training studies is depicted in Figure 1.
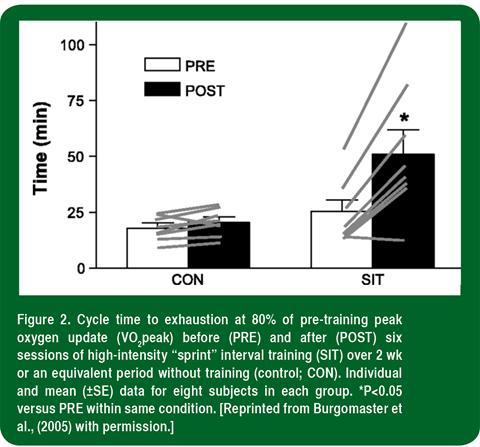
One of the most striking findings from low-volume HIIT studies is the dramatic improvement in exercise capacity during tasks that rely mainly on aerobic energy metabolism, despite the relatively small amount of total exercise training performed (Burgomaster et al., 2005; Gibala et al., 2006; Little et al., 2010). For example, Burgomaster et al. (2005) found that subjects doubled the length of time that exercise could be maintained at a fixed submaximal workload — from ~26 to 51 min during cycling at 80% of pre-training VO2peak — after only six sessions of Wingate-based HIIT over 2 wk (Figure 2).
The validity of this finding was bolstered by the fact that a control group showed no change in performance when tested 2 wk apart with no training intervention. Subsequent work confirmed that the same HIIT protocol improved performance during tasks that more closely resemble normal athletic competition, including laboratory time trials that simulated cycling races lasting from <2 min to <1 h (Gibala et al., 2006). Obviously, the factors responsible for training-induced improvements in exercise capacity are complex and determined by numerous factors including both physiological (e.g., cardiovascular, ionic, metabolic, neural, respiratory) and psychological attributes (e.g., mood, motivation, perception of effort). The short-term studies cited above (Burgomaster et al., 2005; Gibala et al., 2006; Little et al., 2010) reported no measurable change in VO2peak after 2 wk of low-volume HIIT, which suggests the improved exercise performance was primarily attributable to peripheral adaptations in skeletal muscle, as considered further below. Some studies have reported improvements in VO2peak after as little as 2 wk of Wingate-based HIIT, although these were generally conducted on previously sedentary, less fit individuals (Whyte et al., 2010). For a comprehensive overview in this regard, the interested reader is referred to several recent reviews including a meta-analysis (Bacon et al., 2013) that have considered VO2peak trainability in response to HIIT.
PHYSIOLOGICAL ADAPTATIONS TO LOW-VOLUME HIIT
Similar to traditional endurance or strength training, physiological adaptations to HIIT are highly dependent on the precise nature of the training stimulus, i.e., the frequency, intensity and volume of work performed. However, unlike the other two general categories of exercise training which primarily rely on either oxidative (endurance) or non-oxidative (strength) energy to fuel ATP provision, the bioenergetics of high-intensity exercise can differ markedly depending on the duration and intensity of each interval, the number of intervals performed and the nature and duration of recovery between efforts (Ross & Leveritt, 2001). For example, during a single 30-s all-out maximal cycling effort, approximately 20% of total energy provision is derived from oxidative metabolism (Parolin et al., 1999). However, if the exercise bout is repeated three times with four min recovery between bouts, the majority of ATP provision during the final bout is derived from oxidative metabolism (Parolin et al., 1999).
The increased contribution from oxidative metabolism during repeated high-intensity efforts is attributable to both an increased rate of oxygen transport and utilization and decreased ability to stimulate substrate phosphorylation through phosphocreatine hydrolysis and glycolysis (Parolin et al., 1999). High-intensity intermittent exercise is therefore unique because cellular energy during an acute bout or a given training session can be derived primarily from non-oxidative or oxidative metabolism. Consequently, HIIT can elicit a broad range of physiological adaptations and the reader is referred elsewhere for a more comprehensive description of physiological adaptations to HIIT (Buchheit & Laursen, 2013ab; Kubukeli et al., 2002; Ross & Leveritt, 2001). The next sections briefly summarize some of the major metabolic and morphological adaptations to HIIT, again with a focus on recent studies that have examined rapid skeletal muscle remodeling after short-term, low-volume HIIT.
Improved “sprint” or high-intensity exercise performance after HIIT is related in part to increases in the maximal activities of enzymes that regulate non-oxidative energy provision (e.g., glycogen phosphorylase, phosphofructokinase), increased muscle buffering capacity and ionic adaptations including increased sodium-potassium ATPase (Na+-K+-ATPase) content and function (Kubukeli et al., 2002; Ross & Leveritt, 2001). In terms of muscle fiber composition, several studies have reported a bidirectional shift to type IIa (I -> IIa <- IIx), similar to the general trend observed after both endurance and strength training, although this is not a universal finding (Kubukeli et al., 2002; Ross & Leveritt, 2001). HIIT does not have a major effect on muscle size, especially compared to heavy resistance training, although a few studies have reported modest but significant hypertrophy of both Type I and Type II fibers after many months of HIIT (Ross & Leveritt, 2001). In this regard, an acute bout of Wingate-based HIIT does not activate signaling pathways within skeletal muscle that are linked to fiber growth/hypertrophy (Gibala et al., 2009).
It has long been recognized that HIIT also has the potential to increase muscle oxidative capacity and performance during tasks that mainly rely on aerobic energy metabolism (Saltin and Gollnick, 1983). MacDougall et al. (1998) provided an example of the potency of Wingate-based HIIT when they reported an increased maximal activity of several mitochondrial enzymes after 7 wk of training protocol in which subjects performed 4-10 intervals per day, three times per week. Until recently, little was known regarding the early time course and minimum volume of training necessary to elicit rapid adaptations in skeletal muscle, or the effect of HIIT on substrate metabolism during tasks that mainly rely on aerobic energy provision. In a series of studies, we examined rapid adaptations in oxidative energy metabolism and exercise capacity after short-term, Wingate-based HIIT as described above (Burgomaster et al., 2005, 2006, 2007; Gibala et al., 2006). The most unique aspect of this work was the very low training volume, equivalent to only <15 min of very intense exercise or <600 kJ of total work. All studies were performed on healthy college-aged men and women who were habitually active but not engaged in any sort of structured training program.
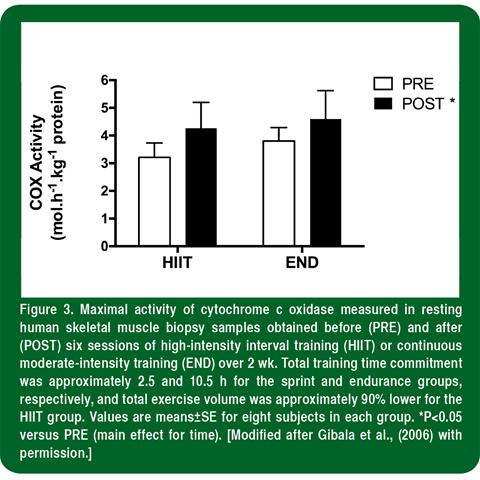
We have consistently found an increased muscle oxidative capacity (assessed using the maximal activity or protein content of mitochondrial enzymes such as citrate synthase and cytochrome oxidase) ranging from <15-35% after six sessions of HIIT over 2 wk (Burgomaster et al., 2005, 2006, 2007). Surprisingly, only a few studies had previously compared changes in muscle oxidative capacity after interval versus continuous training in humans, with equivocal results (see references in Gibala et al., 2006). Moreover, the studies that examined muscle oxidative capacity after interval versus continuous exercise training had used a matched-work design in which total work was similar between groups. In our studies, we sought to compare changes in muscle oxidative capacity and exercise performance after low-volume sprint training and traditional high-volume endurance training, such that the two protocols differed markedly in terms of total training volume and time commitment. The sprint protocol consisted of six sessions of brief, repeated “all-out” 30-s cycling efforts, interspersed with a short recovery, over 14 d. The endurance protocol consisted of six sessions of 90–120 min of moderate intensity cycling exercise, with 1–2 d of recovery interspersed between training sessions. As a result, subjects in both groups performed the same number of training sessions on the same days with the same number of recovery days; however, total training time commitment was 2.5 and 10.5 h, respectively, for the sprint and endurance group, and training volume differed by 90% (630 versus 6500 kJ). The two diverse training protocols induced remarkably similar adaptations in exercise performance and skeletal muscle oxidative capacity, as reflected by the maximal activity of cytochrome c oxidase (Figure 3).
In addition to an increased skeletal muscle oxidative capacity after 2 wk of HIIT, we have also detected changes in carbohydrate metabolism that are normally associated with traditional endurance training, including an increased resting glycogen content and reduced rate of glycogen utilization during matched-work exercise (Burgomaster et al., 2006, 2007). Selected markers of fatty acid metabolism, including the maximal activity of β-hydroxyacyl- CoA dehydrogenase (HAD) and the muscle contents of fatty acid translocase (FAT/CD36) or plasma membrane associated fatty acid binding protein (FABPpm), were unchanged after our short-term Wingate-based training intervention (Burgomaster et al., 2006, 2007), although we have demonstrated an increased HAD after 6 wk of this type of training. Talanian and co-workers (2007) showed that seven sessions of HIIT over 2 wk increased the maximal activity of HAD, the muscle protein content of FABPpm and whole-body fat oxidation during 60 min of cycling at 65% pre-training VO2peak.
A major discrepancy between the respective 2-wk training protocols was the nature of the HIIT stimulus. Subjects did not perform all-out sprints in the study by Talanian et al. (2007), however each training session consisted of 10 x 4-min bouts of cycling at ~90% of VO2peak with 2 min of rest between intervals. Total training time commitment (~5 h) and exercise volume (~3000 kJ) over the 2-wk training period was thus substantially higher than in the studies that employed Wingate-based exercise training (Burgomaster et al., 2006, 2007).
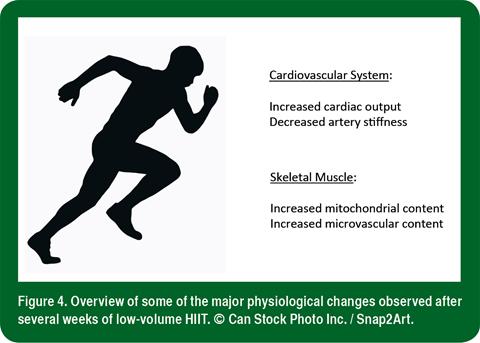
With respect to cardiovascular adaptations, 8 wk of low-volume HIIT has been reported to increase both left ventricular mass and stroke volume (Matsuo et al., 2014). Similar improvements in peripheral vascular structure and function, including popliteal artery distensibility and flow-mediated dilation, were reported after 6 wk of Wingate-based HIIT and traditional moderate-intensity continuous training (Rakobowchuk et al., 2008). A study from a different laboratory that employed the same experimental protocol showed similar improvements in skeletal muscle microvascular density and microvascular enzyme content, despite large differences in total training volume (Cocks et al., 2013). An overview of some of the major physiological adaptations to low-volume HIIT is summarized in Figure 4.
HOW DOES HIIT STIMULATE ADAPTATIONS IN SKELETAL MUSCLE?
The potency of HIIT to elicit rapid changes in skeletal muscle remodeling is no doubt related to its high level of muscle fiber recruitment and potential to stress type II fibers in particular (Saltin & Gollnick, 1983), but the underlying mechanisms are unclear. From a cell-signaling perspective, exercise is typically classified as either “strength” or “endurance,” with short-duration, high-intensity work usually associated with increased skeletal muscle mass, and prolonged, low- to moderate-intensity work associated with increased mitochondrial mass and oxidative enzyme activity (Baar, 2006). Indeed, the distinct pathways that regulate either cell growth or mitochondrial biogenesis intersect at a number of points in an inhibitory fashion, resulting in a response that is largely exclusive for one type of exercise or the other (Baar, 2006). Until recently, little was known regarding the intracellular signaling events that mediate skeletal muscle remodeling in response to HIIT which, unlike traditional strength training, is not characterized by marked skeletal muscle hypertrophy (Ross & Leveritt, 2001).
Given the oxidative phenotype that is rapidly upregulated by HIIT, it seems likely that metabolic adaptations to this type of exercise could be mediated in part through signaling pathways normally associated with endurance training. Contraction-induced metabolic disturbances activate several kinases and phosphatases involved in signal transduction, including the AMP-activated protein kinase (AMPK) and mitogen-activated protein kinase (MAPK) cascades. These signaling pathways have been shown to play a role in promoting specific coactivators involved in mitochondrial biogenesis and metabolism, including activation of peroxisome-proliferator activated receptor γ coactivator (PGC)-1α, which is regarded as the “master regulator” of mitochondrial biogenesis in muscle (Coffee & Hawley, 2007).
It has been demonstrated that Wingate-based HIIT acutely stimulates markers of AMPK and MAPK signaling and increases PGC-1α mRNA by several fold (Gibala et al., 2009; Little et al., 2011), similar to what has been reported after continuous moderate-intensity exercise (Little et al., 2010). Also similar to traditional endurance exercise, acute Wingate-based HIIT may activate PGC-1α by increasing its nuclear translocation (Little et al., 2011), and several weeks of HIIT leads to increased PGC-1α protein content (Burgomaster et al., 2008), suggesting that PGC-1α is likely involved in regulating some of the metabolic adaptations to this form of training. There is also evidence to show that repeated, transient increases in mRNA in response to successive bouts of HIIT lead to sustained increases in the content of transcription and metabolic proteins, eventually resulting in greater mitochondrial protein content and enzyme activity (Perry et al., 2010).
PRACTICAL APPLICATIONS
- HIIT is often dismissed as being only for elite athletes. However, the basic concept of alternating high- and low-intensity periods of exercise can be applied to almost any level of initial fitness.
- As with any form of physical activity, there are potential benefits as well as certain limitations associated with interval training.
- The “pros” include the fact that intervals are a potent training stimulus, and even though the total amount of exercise performed can be quite small, the training can induce adaptations similar to those associated with more prolonged period of continuous moderate-intensity exercise, which makes it relatively time-efficient.
- The “cons” include the uncomfortable feeling of HIIT owing to the relatively intense effort that is required in order to make the training time-efficient, and also the potential greater risk of injury especially if running is performed as compared to less weight-bearing activities such as cycling or swimming.
- While HIIT is effective to improve fitness, there is no magic formula or “one size fits all” approach that is best for everyone, and indeed the best long-term approach is a varied strategy that incorporates strength, endurance and speed sessions as well as flexibility exercises and proper nutrition.
SUMMARY
Highly-trained endurance athletes have long appreciated the role for HIIT as part of a comprehensive training program. Recent evidence shows that — in young healthy persons of average fitness — intense interval exercise is a time-efficient strategy to stimulate skeletal muscle adaptations comparable to traditional endurance training. As little as six sessions of HIIT over 2 wk, or a total of only <15 min of very intense exercise, can increase skeletal muscle oxidative capacity and improve performance during tasks that rely mainly on aerobic energy metabolism. These findings should not be interpreted to suggest that low-volume HIIT provides all of the benefits normally associated with traditional endurance training. The duration of the training programs in the published work to date is relatively short (i.e., lasting up to several weeks) and it remains to be determined whether similar adaptations are manifest after many months of low-volume interval and high-volume continuous training. It is possible that the time course for physiological adjustments differs between training protocols; the very intense nature of interval training may stimulate rapid changes, whereas the adaptations induced by traditional endurance training may occur more slowly. From an applied practical perspective, it will also be important in future work to identify the optimal combination(s) of training intensity and volume necessary to induce adaptations in a time-efficient manner, given that lack of time remains the most commonly cited barrier to lack of regular exercise participation.
REFERENCES
Baar K. (2006). Training for endurance and strength: lessons from cell signaling. Med. Sci. Sports Exerc. 38:1939-1944.
Bacon, A.P., R.E. Carter, E.A. Ogle, and M.J. Joyner (2013). VO2max trainability and high intensity interval training in humans: a meta-analysis. PLoS One. 8:e73182.
Buchheit, M., and P.B. Laursen (2013a). High-intensity interval training, solutions to the programming puzzle: part I: cardiopulmonary emphasis. Sports Med. 43:313-338.
Buchheit, M., and P.B. Laursen (2013b). High-intensity interval training, solutions to the programming puzzle: part II: anaerobic energy, neuromuscular load and practical applications. Sports Med. 43:927-954.
Burgomaster, K.A., N. Cermak, S.M. Phillips, C. Benton, A. Bonen, and M.J. Gibala (2007). Divergent response of metabolite transport proteins in human skeletal muscle after sprint interval training and detraining. Am. J. Physiol. 292:R1970-R1976.
Burgomaster, K.A., G.J.F. Heigenhauser, and M.J. Gibala (2006). Effect of short-term sprint interval training on human skeletal muscle carbohydrate metabolism during exercise and time trial performance. J. Appl. Physiol. 100:2041-2047.
Burgomaster, K.A., K.R. Howarth, M. Rakobowchuk, S.M. Phillips, M.J. MacDonald, S. McGee, and M.J. Gibala (2008). Similar metabolic adaptations during exercise after low volume sprint interval and traditional endurance training in humans. J. Physiol. 586:151-160.
Burgomaster, K.A., S.C. Hughes, G.J.F. Heigenhauser, S.N. Bradwell, and M.J. Gibala (2005). Six sessions of sprint interval training increases muscle oxidative potential and cycle endurance capacity. J. Appl. Physiol. 98:1895- 1990.
Cocks, M., C.S. Shaw, S.O. Shepherd, J.P. Fisher, A.M. Ranasinghe, T.A. Barker, K.D. Tipton, and A.J. Wagenmakers. (2013). Sprint interval and endurance training are equally effective in increasing muscle microvascular density and eNOS content in sedentary males. J. Physiol. 591:641-656.
Coffey, V.G., and J.A. Hawley (2007). The molecular bases of training adaptation. Sports Med. 37:737-763.
Gibala M.J., J.B. Gillen J.B., and M.E. Percival (2014). Physiological and health-related adaptations to low-volume interval training: influences of nutrition and sex. Sports Med. 44 Suppl 2:127-137.
Gibala, M.J., J.P. Little, M. van Essen, G.P. Wilkin, K.A. Burgomaster, A. Safdar, S. Raha, and M.A.Tarnopolsky (2006). Short-term sprint interval versus traditional endurance training: similar initial adaptations in human skeletal muscle and exercise performance. J. Physiol. 575:901-911.
Gibala, M.J., J.P. Little, M.J. MacDonald, and J.A. Hawley (2012). Physiological adaptations to low volume, high-intensity interval training in health and disease. J. Physiol. 590:1077-1084.
Gibala, M.J., S.L. McGee, A. Garnham, K. Howlett, R. Snow, and M. Hargreaves M (2009). Brief intense interval exercise activates AMPK and p38 MAPK signaling and increases the expression of PGC-1α in human skeletal muscle. J. Appl. Physiol. 106:929-934.
Kessler H.S., S.B. Sisson, and K.R. Short (2012). The potential for high-intensity interval training to reduce cardiometabolic disease risk. Sports Med. 42:489-509.
Kubukeli, Z.N., T.D. Noakes, and S.C. Dennis (2002). Training techniques to improve endurance exercise performances. Sports Med. 32:489-509.
Laursen, P.B., and D.G. Jenkins (2002). The scientific basis for high-intensity interval training: optimising training programmes and maximising performance in highly trained endurance athletes. Sports Med. 32:53-73.
Little, J.P., A. Safdar, D. Bishop, M.A. Tarnopolsky, and M.J. Gibala (2011). An acute bout of high-intensity interval training increases the nuclear abundance of PGC- 1α and activates mitochondrial biogenesis in human skeletal muscle. Am. J. Physiol. 300:R1303-1310.
Little, J.P., A.S. Safdar, G.P. Wilkin, M.A. Tarnopolsky, and M.J. Gibala (2010). A practical model of low-volume high-intensity interval training induces mitochondrial biogenesis in human skeletal muscle: potential mechanisms. J. Physiol. 586:1011-1022.
MacDougall, J.D., A.L. Hicks, J.R. MacDonald, R.S. McKelvie, H.J. Green, and K.M. Smith (1998). Muscle performance and enzymatic adaptations to sprint interval training. J. Appl. Physiol. 84:2138-2142.
Matsuo, T., K. Saotome, S. Seino, N. Shimojo, A. Matsushita, M. Iemitsu, H. Ohshima, K. Tanaka, and C. Mukai C. (2014). Effects of a low-volume aerobic-type interval exercise on VO2max and cardiac mass. Med. Sci. Sports Exerc. 46:42-50.
Parolin, M.L., A. Chesley, M.P. Matsos, L.L. Spriet, N.L. Jones, and G.J.F. Heigenhauser (1999). Regulation of skeletal muscle glycogen phosphorylase and PDH during maximal intermittent exercise. Am. J. Physiol. 277:E890-900.
Perry, C.G., J. Lally, G.P. Holloway, G.J. Heigenhauser, A. Bonen, and L.L. Spriet LL (2010). Repeated transient mRNA bursts precede increases in transcriptional and mitochondrial proteins during training in human skeletal muscle. J. Physiol. 588:4795-4810.
Rakobowchuk, M. S. Tanguay, K.A. Burgomaster, K.R. Howarth, M.J. Gibala, and M.J. MacDonald. Sprint interval and traditional endurance training induce similar improvements in peripheral arterial stiffness and flow-mediated dilation in healthy humans. Am. J. Physiol. 295:R236-R242, 2008.
Ross A., and M. Leveritt (2001). Long-term metabolic and skeletal muscle adaptations to short-sprint training: implications for sprint training and tapering. Sports Med. 31:1063-1082.
Saltin, B., and P.D. Gollnick (1983). Skeletal muscle adaptability: significance for metabolism and performance. In L.D. Peachey (ed.) Handbook of Physiology. Skeletal Muscle. pp. 555-631, American Physiological Society, Bethesda.
Talanian, J.L., S.D. Galloway, G.J.F. Heigenhauser, A. Bonen, and L.L. Spriet (2007). Two weeks of high-intensity aerobic interval training increases the capacity for fat oxidation during exercise in women. J. Appl. Physiol. 102:1439-1447.
Weston, K.S., U. Wisløff, and J.S. Coombes. High-intensity interval training in patients with lifestyle-induced cardiometabolic disease: a systematic review and meta-analysis. Br. J. Sports Med. 48:1227-1234, 2014.
Whyte, L.J., J.M. Gill, and A.J. Cathcart (2010). Effect of 2 weeks of sprint interval training on health-related outcomes in sedentary overweight/obese men. Metabolism. 59:1421-1428.








