MINIMIZING INJURY AND MAXIMIZING RETURN TO PLAY RESEARCH: Lessons from Engineered Ligaments

KEY POINTS
- Soft tissue injuries account for upwards of 70% of injuries during sports. One of the reasons for this high injury rate is that we have only a very basic understanding of how training alters tendon and ligament structure and function.
- Tendons and ligaments are complex and dynamic tissues that are poorly understood, in part because of the difficulty in working with them in human models (in vivo).
- Because of the difficulty in measuring function and imaging effectively in vivo, it would be useful to have an outside the body or in vitromodel that can fully recapitulate the tissue.
- Recently, engineered ligaments have been developed in vitrofrom fibroblasts isolated from human anterior cruciate ligaments or hamstring tendons. These tissues are functionally similar to embryonic tendons and ligaments and can be used to understand how different stimuli affect sinew function.
- Using these ligaments, research has shown that tendons and ligaments, like bone, quickly become refractory to an exercise stimulus, suggesting that short (10 min) periods of activity are best to train these tissues.
- These tissues are also being used to understand how hormonal changes (menstrual cycle or intense exercise) and nutritional interventions can affect sinew function.
- The challenge to tissue engineering will be to translate these novel insights into new nutritional and training regimes to minimize injury and accelerate return to play.
INCIDENCE OF SOFT TISSUE INJURY IN SPORT
Soft tissue injuries that affect muscles, tendons and ligaments are extremely common at all levels of sport. In youth sport, ~50% of all injuries are sprains (Frisch et al., 2009). In college athletics, the rates of ankle sprains have been relatively constant at ~1/1,000 athlete exposures, over the 15 years between 1988 and 2004 (Hootman et al., 2007). By contrast, anterior cruciate ligament (ACL) ruptures increased by 1.3%/yr to 0.14/1,000 athlete exposures over the same time period. In professional sports, the incidence of soft tissue injury reaches 60% for the English Premier League (Hawkins et al., 2001) and nearly 70% in the National Football League (Feeley et al., 2008). Beyond sporting populations, diabetics are up to 10 times more likely to suffer tendon injuries than non-diabetics (Abate et al., 2013)and tendon injuries increase with age as evidenced by the fact that 80% of people in their 80s have suffered a ruptured tendon (Milgrom et al., 1995). Even though musculoskeletal injuries are extremely common and have huge personal, competitive and financial costs, very few advances have been made in preventing and treating these injuries.
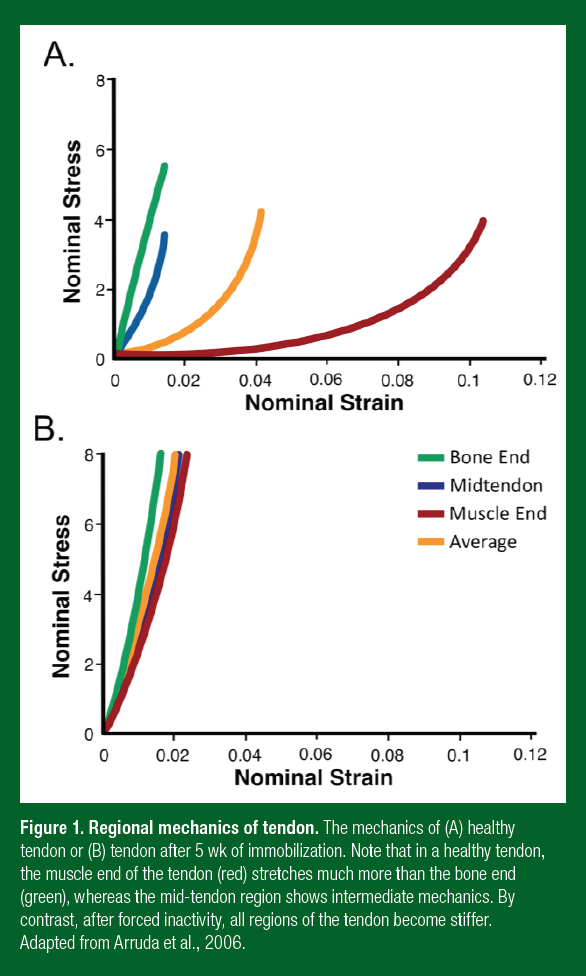
Tendons and ligaments are often grouped together into a single category (the biblical term “sinew” will be used in this review) because they are similar in their composition, structure and general function. The fundamental difference between these tissues is that a tendon attaches a compliant muscle to a stiff bone, whereas a ligament attaches two stiff bones together. The magnitude of this difference is often underappreciated. Connecting a compliant muscle to a stiff bone can cause strain concentrations (where one tissue stretches much more than the other). When the strain concentration gets too high, failure or rupture occurs at the interface or within the compliant tissue. The tendon prevents strain concentrations by having variable mechanics along its length (Arruda et al., 2006) (Figure 1). This means that a healthy tendon starts stretchy at the muscle end and becomes progressively stiffer as it gets closer to the bone. The compliant region of tendon is believed to protect the attached muscle from injury, by acting as a shock absorber (minimizing strain concentrations). For example, during running, a healthy tendon lengthens to absorb energy allowing the muscle to contract isometrically (Roberts et al., 1997). However, when stiffness of the muscle end of the tendon is greater than the isometric strength of the muscle, the muscle must lengthen during the movement. This type of rapid lengthening of a contracting muscle can lead to severe injury. This means that even though the strength (load at which they fail) of a tendon or ligament increases as stiffness increases (LaCroix et al., 2013), a stiffer tendon produces more muscle damage during the same exercise than a less stiff tendon (McHugh et al., 1999). For an athlete, this means that the stiffer a ligament the better, but with tendons, too much stiffness can be bad for the attached muscle.
The muscle-end of a tendon stretches more because of differences in the orientation and crosslinking of the collagen within this region. Within a sinew, molecular connections, or crosslinks, bind collagen molecules and fibrils together and increase the stiffness of the tissue (Reddy et al., 2002). These crosslinks are low at the muscle end of a tendon and increase toward the bone (Curwin et al., 1994). Since there are fewer crosslinks at the muscle end, there is lower stiffness in this region (Arruda et al., 2006). There are three ways that we can increase crosslinks and therefore the stiffness at the muscle end of a tendon: 1) high blood sugar – sugar can directly crosslink collagen and this is why diabetics are up to 10 times more likely to get a musculoskeletal injury; 2) inactivity (as in a cast), as without muscle contraction, tendons of the immobilized joint add crosslinks at the muscle end and become stiffer (Arruda et al., 2006; Eliasson et al., 2007); and 3) exercise, which can either increase or decrease crosslinks depending on the speed of the movement. High-speed movements (think plyometrics) increase crosslinks and stiffness, whereas slow movements (think slow eccentrics or isometrics) break crosslinks and lower stiffness.
From the background above, the most important points to take away are: 1) tendon/ligament injuries are common in daily activities and at every level of athletic performance; 2) a healthy tendon has varying stiffness along its length and the compliant region acts as a shock absorber that protects the muscle from injury; and 3) as a result of the tissues they attach to, tendons and ligaments are functionally different. The stiffer the ligament the better, whereas if tendon stiffness is too high, injuries will increase to the associated muscles. This means that training and nutrition that increase sinew stiffness are good, but to prevent tendon/muscle injury slow, heavy movements (i.e., weight lifting) should also be performed. This Sports Science Exchange article will provide a brief overview of how pioneering work is increasing the knowledge regarding the structure and function of tendons and ligaments (see Baar, 2017 for more detail).
MODELING SINEW PHYSIOLOGY IN VITRO
For years, scientists, coaches and athletes considered tendons and ligaments mechanical bands that did not respond to exercise. However, it is now clear that these tissues respond to loading. For example, the patellar tendon in the dominant leg of fencers and badminton players is 20-30% larger than their trail leg (Couppe et al., 2008). Further, nutritional interventions, such as consuming a whey protein supplement, have the potential to increase the tendon hypertrophy that results from strength training (Farup et al., 2014). This means that sinews are dynamic tissues. Interestingly, the existing data suggest that tendons rapidly respond to changes in muscle size and strength, possibly to minimize changes in the peak strain (a stronger muscle can pull with greater force on a tendon and a larger tendon is needed to handle the higher force) during contraction (Urlando & Hawkins, 2007). However, it is important to note that even though part of the tissue is dynamic, the collagen at the core of a tendon does not turn over between ages 17-70 (Heinemeier et al., 2013). Together, these data suggest that an adult tendon grows like a tree, adding and removing collagen only on the outside (Kalliokoski et al., 2007).
Since tendons are dynamic, understanding how exercise and feeding increase the synthesis of collagen and improve tendon function could allow us to improve performance, prevent injury, and accelerate return to play. However, unlike many other tissues in the body, tendons are largely composed of extracellular proteins. In fact, the number of cells within a tendon drops with age until there are fewer than 0.01 cells/micron squared in the adult tendon (Nakagawa et al., 1994). Contrast this with skeletal muscle where ~95% of the area is composed of cells. Therefore, getting enough intracellular protein from a tendon biopsy for scientific experiments can be extremely challenging. Together with the fact that proteins within the core of the tendon turn over very slowly (Heinemeier et al. 2013), means that it is very difficult to understand how the cells within a healthy adult tendon respond to nutrition or exercise.
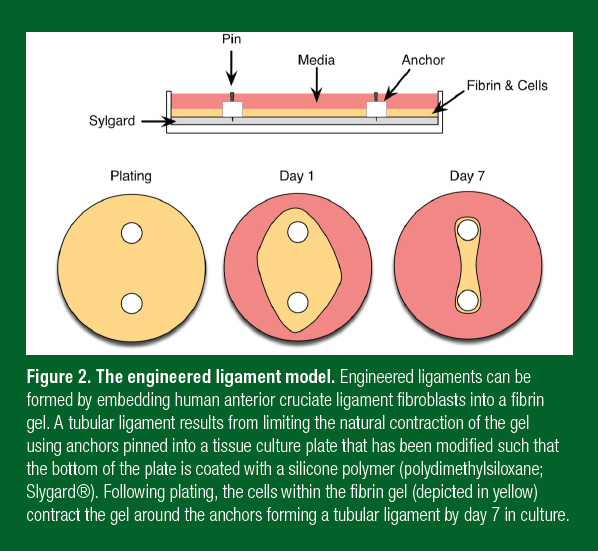
For these reasons, over the last 10 years, a number of laboratories have developed 3-dimensional models of a human tendon/ligament (Bayer et al., 2010; Kapacee et al., 2008; Paxton et al., 2009; 2010). To achieve this goal, our laboratory isolates human fibroblasts from ruptured ACLs that are collected during reconstructive surgery (Paxton et al., 2012). Enzymes are used to digest the collagen, cells are collected as they are released, and the cells are grown in an incubator at body temperature in a media that promotes growth. The cells can be expanded in the lab and this allows hundreds of ligaments to be made from a single donor, eliminating the effects of genetic differences between subjects. To engineer ligaments, cells are put into a gel made from fibrin, the same protein that forms a blood clot when we cut ourselves. This matrix is used because it is the biological matrix that the cells are exposed to in development and injury repair (Galloway et al., 2013). The cells grow and divide within the fibrin gel and over 7 d contract the gel around two calcium phosphate cement anchors that are placed in the culture dish to act like bones (Figure 2). After 7 d, the fibrin contracts into a single tissue between the anchors and continues to develop like an embryonic tendon or ligament (Kapacee et al., 2008). Like developing sinews (Marturano et al., 2013), these engineered tissues have more cells and less matrix (Calve et al., 2010), their rate of collagen synthesis is significantly higher (Calve et al., 2010), they express more developmental collagen isoforms (Bayer et al., 2010), and are much weaker than the adult tissues they are designed to mimic (Paxton et al., 2010). In spite of these significant differences, the engineered tissues provide a model that has been useful in understanding the effects of exercise and nutrition on tendon/ligament function. The following section discusses some
of the exciting findings using this model, how these data compare to the animal/human data (where possible), and how the information derived from this model could potentially improve performance, decrease injuries and accelerate return to play.
LOADING AND SINEW FUNCTION
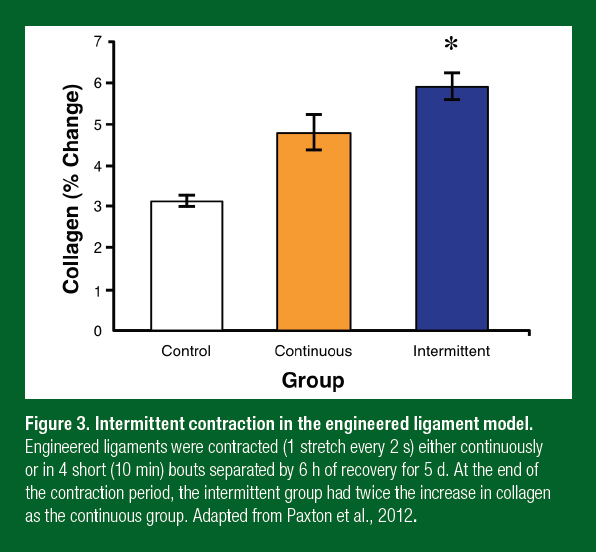
Sinews, like other musculoskeletal tissues such as bone, adapt to their loading state. In the sinews of adult animals and humans, disuse leads to a decrease in total collagen (Loitz et al., 1989; Vailas et al., 1988), whereas exercise increases collagen (Heinemeier et al., 2007). Even though activity is known to regulate cellular processes within tendons and ligaments, how the volume, intensity and load of exercise affects sinews is largely unstudied. To begin to understand what type of exercise was optimal for sinews, we stretched our engineered ligaments at different frequencies, intensities and durations and determined the molecular response (Paxton et al., 2012). These experiments showed that the molecular response to exercise did not depend on the frequency (how many times a second it was loaded; think walking versus running) and intensity (how large the stretch was; think range of motion). This is consistent with animal experiments where the molecular response to resistance exercise was the same in tendons regardless of whether the muscle underwent shortening, isometric, or lengthening contractions (Heinemeier et al., 2007). The only parameter that altered the cellular response to contraction or exercise was time. Within 10 min of starting the activity, the molecular response peaked. If contractions continued, the molecular signals switched off (Paxton et al., 2012). Further experiments showed that it took 6 h for the cells to become responsive to contractions again. Using this information, we developed an intermittent contraction (exercise) program consisting of 10 min of activity followed by 6 h of rest (Paxton et al., 2012). After 5 d, the engineered ligaments that had undergone the intermittent activity protocol produced twice as much collagen as those that were exercised continuously (Figure 3). These results are similar to what occurs in bone in vivo; where very few loading events followed by 6-8 h of rest resulted in the greatest amount of bone mineral deposition (Burr et al., 2002). For athletes and coaches, these data suggest that: repeated short periods of activity that load the sinew followed by long periods of rest appear to be optimum for connective tissue health and function.
HORMONAL EFFECTS ON SINEW FUNCTION
Female athletes participating in cutting and jumping sports have a 4-6 times greater incidence of ACL rupture than their male counterparts (Arendt & Dick, 1995). Interestingly, knee laxity (Shultz et al., 2004; 2005) and incidence of ACL rupture (Wojtys et al., 1998) are related to circulating estrogen levels. As estrogen rises around ovulation, knee laxity and the incidence of ACL rupture both go up as well (Shultz et al., 2005; 2005; Wojtys et al., 1998). This suggests that even though there are well-established biomechanical differences between men and women, hormone levels might directly affect ligament function. To attempt to understand the mechanism underlying this effect, we mimicked the estrogen surge that occurs just before ovulation by increasing estrogen levels in our culture model and measured the changes in mechanics (Lee et al., 2015). Interestingly, as little as 48 h in physiologically high estrogen was enough to decrease the stiffness of our ligaments without changing collagen content. A decrease in stiffness without a change in collagen suggested that there was a decrease in crosslinking. To directly test this hypothesis, we treated our ligaments with high estrogen for 24 or 48 h and measured the activity of lysyl oxidase, the primary collagen crosslinking enzyme. Consistent with our hypothesis, estrogen decreased the activity of lysyl oxidase by more than 80% (Lee et al., 2015). This indicated that the increase in estrogen during the menstrual cycle decreased crosslinking resulting in decreased stiffness that put the ACL at a greater risk of failure. We are now using our engineered ligament model to search for nutritional supplements that would have the opposite effect in an effort to decrease ACL injury in athletic women.
While estrogen is known to decrease sinew function, as described above exercise has a positive effect. While most of the benefit of exercise is a direct effect of loading (Farup et al., 2014; Paxton et al., 2012), exercise is known to have a globally positive effect on connective tissues as well (Crane et al., 2015). To test whether the hormonal changes that result from strength training are beneficial for sinew function, we sampled blood from 12 healthy young men before and after resistance exercise (West et al., 2015). The serum from these subjects was isolated and then used in the growth media of our engineered ligaments. We then determined the effect of the different sera on collagen synthesis and mechanics. Constructs grown in the post exercise serum showed a significant increase in collagen content and mechanics, suggesting that something in the post exercise serum improved sinew function (West et al., 2015). This effect was not mediated by growth hormone, transforming growth factor b, or insulin-like growth factor (IGF) 1, but to date we have not been unable to discover the beneficial factor. These data suggest that exercise produces a global signal that improves connective tissue function. Identifying this factor might provide a way to improve sinew function and health, and accelerate return to play. In the meantime, these data suggest that even before you can load an injured sinew, engaging in heavy exercise with healthy body parts might produce factors that could accelerate recovery.
NUTRITIONAL INTERVENTIONS TO IMPROVE SOFT TISSUE FUNCTION
Compared to muscle, the science of nutritional interventions that can improve soft tissue function in humans is in its infancy. One paper showed that whey protein improved tendon hypertrophy in response to strength training (Farup et al., 2014). However, whether this was a direct effect on the tendon or an indirect effect resulting from more muscle hypertrophy and strength gains is still unclear. Interestingly, a similar response was seen in rats following 5 wk of leucine supplementation (Barbosa et al., 2012). Both whey and leucine work through the mechanistic target of rapamycin (mTORC1) (Wolfson et al., 2016). To determine whether mTORC1 activity was important in collagen synthesis and sinew mechanics, we added rapamycin to our culture media to block mTORC1. Treatment with rapamycin for 7 d decreased the mechanics and collagen content of the grafts by more than 50% (Baar, unpublished observations). However, whether these data are the result of a decrease in collagen synthesis, or a decrease in cell number has yet to be determined. Regardless, these data suggest that using leucine-rich whey protein can activate mTORC1 within sinews and increase collagen. However, the effects of whey protein on sinew structure and mechanics in humans has yet to be determined.
Other than leucine-rich protein, no nutritional interventions have been shown to have an effect on sinew in humans. However, using our tissue engineered ligament model we have shown that certain amino acids together with vitamin C can improve collagen synthesis (Paxton et al., 2010). The vitamin C effect is not surprising given that vitamin C deficiency results in scurvy, a disease marked by the loss of collagen synthesis (Peterkofsky, 1991). Vitamin C is an essential co-factor for an enzyme that is needed for the synthesis, secretion and crosslinking of collagen. In our model, the amino acids that have a positive effect on collagen synthesis include proline, lysine, hydroxylysine and hydroxyproline. These amino acids are the main components of collagen, suggesting that even in our in vitromodel, where amino acids are 5 times the physiological level, excess proline, lysine and their hydroxylated analogues can still be beneficial. Interestingly, these same amino acids are enriched in hydrolyzed collagen and gelatin, which is usually made from the tendons and ligaments of cows. In agreement with our findings in engineered sinews, in a randomized clinical trial, Clark and colleagues (2008) showed that hydrolyzed collagen decreased knee pain in athletes. Further, McAlindon and colleagues (2011) showed that consuming 10 g/d of collagen hydrolysate resulted in an increase in cartilage within the knee. These findings suggest that gelatin or hydrolyzed collagen have important benefits for athletes; however, whether this is the result of more collagen synthesis remains to be determined. We have begun feeding gelatin to athletes and have seen extremely positive responses on collagen production (Shaw et al., 2017). In this randomized double-blind crossover designed clinical trial we have seen that consuming 15 g of gelatin 1 h before activity resulted in a significant increase in collagen synthesis in people (Shaw et al., 2017). Further, engineered ligaments grown in serum after ingesting the vitamin C enriched collagen supplement resulted in a dose-dependent increase in collagen and an improvement in mechanics. These data suggest that consuming gelatin improves tendon collagen synthesis in people; however, more work is needed to understand the dose and the specificity of this response to collagen (i.e., can any protein improve collagen synthesis?).
SCIENCE-BASED RECOMMENDATIONS FOR TRAINING TO IMPROVE TENDON HEALTH AND PERFORMANCE
From the background provided above, a series of recommendations can be developed to help maximize performance, decrease the risk of tendon/ligament injury and/or accelerate return to play.
- In healthy athletes with a history of tendon injuries, consider incorporating a connective tissue health session into training. This type of session would involve ~10 min of activity targeted to the tendon/ligament that is prone to injury. For example, runners would do a session to target the hamstrings, patellar and Achilles tendons, whereas baseball players would target the throwing arm. These exercises can be performed at high or low speed, with a lighter weight and with a limited range of motion if necessary since the only goal is to go for ~10 min to stimulate the cells to adapt, and the exercises should be performed either 6 h before or after any other training.
- In healthy athletes with a history of muscle pulls, consider incorporating heavy slow strength training to decrease crosslinks and stiffness in the muscle end of the tendon. These sessions can use concentric, isometric or eccentric loading; the key to decrease muscle injuries is the slow speed of the movement which will decrease stiffness at the muscle end of the tendon. Again, these can be short sessions ~10 min that can follow immediately at the end of a game or practice.
- Following injury, athletes should begin training as soon as possible. Training can consist of simple isometric contractions, unweighted range of motion, and weight-supported exercises since the load and amplitude of the movement is not important for stimulating repair (Paxton et al., 2012). The training should again consist of 10 min of activity followed by 6 h of rest. Reasonably, this means that the athlete will train for three short periods each day: in the morning, at noon and in the evening. Heavy weight training of uninjured areas may also provide other beneficial factors that could accelerate recovery (West et al., 2015).
- Consume leucine-rich protein as part of your training. Beyond the direct effects on muscle (Churchward-Venne et al., 2012), tendon may also benefit from the added muscle mass and strength and possibly greater mTORC1 activation (Farup et al., 2014).
- Blood flow to inactive tendon is very low and therefore, nutrient delivery to tendon at rest is believed to be limited. Glucose uptake into tendon increases during exercise (Bojsen-Moller et al., 2006). This suggests that tendons increase their metabolic activity during exercise and therefore any nutritional intervention designed to directly target a tendon/ligament may work better if supplied before exercise.
- Our initial work suggests that ~60 min before training, athletes should be encouraged to consume ~15 g of gelatin in either liquid or gel form (Shaw et al., 2017). The exact amount of gelatin and whether this will vary with body weight is currently being investigated.
Figure legends
Figure 1. Regional mechanics of tendon. The mechanics of (A) healthy tendon or (B) tendon after 5 wk of immobilization. Note that in a healthy tendon, the muscle end of the tendon (red) stretches much more than the bone end (green), whereas the mid-tendon region shows intermediate mechanics. By contrast, after forced inactivity, all regions of the tendon become stiffer. Adapted from Arruda et al., 2006.
Figure 2. The engineered ligament model. Engineered ligaments can be formed by embedding human anterior cruciate ligament fibroblasts into a fibrin gel. A tubular ligament results from limiting the natural contraction of the gel using anchors pinned into a tissue culture plate that has been modified such that the bottom of the plate is coated with a silicone polymer (polydimethylsiloxane; Slygard®). Following plating, the cells within the fibrin gel (depicted in yellow) contract the gel around the anchors forming a tubular ligament by day 7 in culture.
Figure 3. Intermittent contraction in the engineered ligament model. Engineered ligaments were contracted (1 stretch every 2 s) either continuously or in 4 short (10 min) bouts separated by 6 h of recovery for 5 d. At the end of the contraction period, the intermittent group had twice the increase in collagen as the continuous group. Adapted from Paxton et al., 2012.
References
Abate, M., C. Schiavone, V. Salini, and I. Andia (2013). Occurrence of tendon pathologies in metabolic disorders. Rheumatology52:599-608.
Arendt, E., and R. Dick (1995). Knee injury patterns among men and women in collegiate basketball and soccer. NCAA data and review of literature. Am. J. Sports Med.23:694-701.
Arruda, E.M., S. Calve, R.G. Dennis, K. Mundy, and K. Baar (2006). Regional variation of tibialis anterior tendon mechanics is lost following denervation. J. Appl. Physiol.101:1113-1117.
Baar, K. (2017). Minimizing injury and maximizing return to play: lessons from engineered ligaments. Sports Med.47:S5-S11.
Barbosa, A.W., G.P. Benevides, L.M. Alferes, E.M. Salomao, M.C. Gomes-Marcondes, and L.A. Gomes (2012). A leucine-rich diet and exercise affect the biomechanical characteristics of the digital flexor tendon in rats after nutritional recovery. Amino Acids42:329-336.
Bayer, M.L., C.Y. Yeung, K.E. Kadler, K. Qvortrup, K. Baar, R.B. Svensson, S.P. Magnusson, M. Krogsgaard, M. Koch, and M. Kjaer (2010). The initiation of embryonic-like collagen fibrillogenesis by adult human tendon fibroblasts when cultured under tension. Biomaterials31:4889-4897.
Bojsen-Moller, J., K.K. Kalliokoski, M. Seppanen, M. Kjaer, and S.P. Magnusson (2006). Low-intensity tensile loading increases intratendinous glucose uptake in the Achilles tendon. J. Appl. Physiol.101:196-201.
Burr, D.B., A.G. Robling, and C.H. Turner (2002). Effects of biomechanical stress on bones in animals. Bone30:781-786.
Calve, S., I.F. Lytle, K. Grosh, A.L. Brown, and E.M. Arruda (2010). Implantation increases tensile strength and collagen content of self-assembled tendon constructs. J. Appl. Physiol.108:875-881.
Churchward-Venne, T.A., N.A. Burd, and S.M. Phillips (2012). Nutritional regulation of muscle protein synthesis with resistance exercise: strategies to enhance anabolism. Nutr. Metab.9:40.
Clark, K.L., W. Sebastianelli, K.R. Flechsenhar, D.F. Aukermann, F. Meza, R.L. Millard RL, J.R. Deitch, P.S. Sherbondy, and A. Albert (2008).24-Week study on the use of collagen hydrolysate as a dietary supplement in athletes with activity-related joint pain. Curr. Med. Res. Opin.24:1485-1496.
Couppe, C., M. Kongsgaard, P. Aagaard, P. Hansen, J. Bojsen-Moller, M. Kjaer, and S.P. Magnusson (2008). Habitual loading results in tendon hypertrophy and increased stiffness of the human patellar tendon. J. Appl. Physiol.105:805-810.
Crane, J.D., L.G. MacNeil, J.S. Lally, R.J. Ford, A.L. Bujak, I.K. Brar, B.E. Kemp, S. Raha, G.R. Steinberg, and M.A. Tarnopolsky (2015).Exercise-stimulated interleukin-15 is controlled by AMPK and regulates skin metabolism and aging. Aging Cell14:625-634.
Curwin, S.L., R.R. Roy, and A.C Vailas (1994). Regional and age variations in growing tendon. J. Morphol.221:309-320.
Eliasson, P., A. Fahlgren, B. Pasternak, and P. Aspenberg P (2007). Unloaded rat achilles tendons continue to grow but lose viscoelasticity. J. Appl. Physiol.103:459-463.
Farup, J., S.K. Rahbek, M.H. Vendelbo, A. Matzon, J. Hindhede, A. Bejder, S. Ringgard, and K. Vissing (2014)Whey protein hydrolysate augments tendon and muscle hypertrophy independent of resistance exercise contraction mode. Scand. J. Med. Sci. Sports24:788-798.
Feeley, B.T., S. Kennelly, R.P. Barnes, M.S. Muller, B.T. Kelly, S.A. Rodeo, and R.F. Warren (2008). Epidemiology of National Football League training camp injuries from 1998 to 2007. Am. J. Sports Med.36:1597-1603.
Frisch, A., J.L. Croisier, A. Urhausen, R. Seil, and D. Theisen (2009). Injuries, risk factors and prevention initiatives in youth sport. Br. Med. Bull.92:95-121.
Galloway, M.T., A.L. Lalley, and J.T. Shearn (2013). The role of mechanical loading in tendon development, maintenance, injury, and repair. J. Bone Joint Surg. Am.95:1620-1628.
Hawkins, R.D., M.A. Hulse, C. Wilkinson, A. Hodson, and M. Gibson (2001). The association football medical research programme: an audit of injuries in professional football. Br. J. Sports Med.35:43-47.
Heinemeier, K.M., J.L. Olesen, F. Haddad, H, Langberg, M. Kjaer, K.M. Baldwin, and P. Schjerling (2007). Expression of collagen and related growth factors in rat tendon and skeletal muscle in response to specific contraction types. J. Physiol.582:1303-1316.
Heinemeier, K.M., P. Schjerling, J. Heinemeier, S.P. Magnusson, and M. Kjaer (2013). Lack of tissue renewal in human adult Achilles tendon is revealed by nuclear bomb 14C. FASEB J.27:2074-2079.
Hootman, J.M., R. Dick, and J. Agel (2007). Epidemiology of collegiate injuries for 15 sports: summary and recommendations for injury prevention initiatives. J, Athl. Train.42:311-319.
Kalliokoski, K.K., J. Bojsen-Moller, M. Seppanen, J. Johansson, M. Kjaer, M. Teras, and S.P. Magnusson (2007).Contraction-induced [18F]-fluoro-deoxy-glucose uptake can be measured in human calf muscle using high-resolution PET. Clin. Physiol. Funct. Imaging27:239-241.
Kapacee, Z., S.H. Richardson, Y. Lu, T. Starborg, D.F. Holmes, K. Baar, and K.E.Kadler (2008). Tension is required for fibripositor formation. Matrix Biol.27:371-375.
LaCroix, A.S., S.E. Duenwald-Kuehl, R.S. Lakes, and R. Vanderby, Jr (2013). Relationship between tendon stiffness and failure: a metaanalysis. J. Appl. Physiol.115:43-51.
Lee. C., A. Lee-Barthel, L. Marquino, N. Sandoval, G. Marcotte, and K. Baar (2015). Estrogen inhibits lysyl oxidase and decreases mechanical function in engineered ligaments. J. Appl. Physiol.118:1250-1257.
Loitz, B.J., R.F. Zernicke, A,C. Vailas, M.H. Kody, and R.A Meals (1989). Effects of short-term immobilization versus continuous passive motion on the biomechanical and biochemical properties of the rabbit tendon. Clin. Orthop.244:265-271.
Marturano, J.E., J.D. Arena, Z.A. Schiller, I. Georgakoudi, and C.K. Kuo CK (2013). Characterization of mechanical and biochemical properties of developing embryonic tendon. Proc. Nat. Acad. Sci. USA. 110:6370-6375.
McAlindon, T.E., M. Nuite, N. Krishnan, R. Ruthazer, L.L. Price, D. Burstein, J. Griffith, and K. Flechsenhar (2011). Change in knee osteoarthritis cartilage detected by delayed gadolinium enhanced magnetic resonance imaging following treatment with collagen hydrolysate: a pilot randomized controlled trial. Osteo. Cart.19:399-405.
McHugh, M.P., D.A. Connolly, R.G. Eston, I.J. Kremenic, S.J. Nicholas, and G.W. Gleim GW (1999). The role of passive muscle stiffness in symptoms of exercise-induced muscle damage. Am. J. Sports Med.27:594-599.
Milgrom, C., M. Schaffler, S. Gilbert, and M. van Holsbeeck (1995). Rotator-cuff changes in asymptomatic adults. The effect of age, hand dominance and gender. J. Bone Joint Surg. Br.77:296-298.
Nakagawa, Y., T. Majima, and K. Nagashima (1994). Effect of ageing on ultrastructure of slow and fast skeletal muscle tendon in rabbit Achilles tendons. Acta Physiol. Scand.152:307-313.
Paxton, J.Z., K. Donnelly, R.P. Keatch, and K. Baar (2009). Engineering the bone-ligament interface using polyethylene glycol diacrylate incorporated with hydroxyapatite. Tissue Eng. Part A.15:1201-1209.
Paxton, J.Z., L.M. Grover, K. Baar (2010). Engineering an in vitro model of a functional ligament from bone to bone. Tissue Eng. Part A.16:3515-3525.
Paxton, J.Z., P. Hagerty, J.J. Andrick, and K. Baar (2012). Optimizing an intermittent stretch paradigm using ERK1/2 phosphorylation results in increased collagen synthesis in engineered ligaments. Tissue Eng. Part A.18:277-284.
Peterkofsky, B. (1991). Ascorbate requirement for hydroxylation and secretion of procollagen: relationship to inhibition of collagen synthesis in scurvy. Am. J. Clin. Nutr.54:1135S-1140S.
Reddy, G.K., L. Stehno-Bittel, and C.S. Enwemeka (2002). Glycation-induced matrix stability in the rabbit achilles tendon. Arch Biochem. Biophys.399:174-180.
Roberts, T.J., R.L. Marsh, P.G. Weyand, and C.R. Taylor (1997). Muscular force in running turkeys: the economy of minimizing work. Science275:1113-1115.
Shaw, G., A. Lee-Barthel, M.L. Ross, B. Wang, and K. Baar (2017). Vitamin C-enriched gelatin supplementation before intermittent activity augments collagen synthesis. Am. J. Clin. Nutr.105:136-143.
Shultz, S.J., S.E. Kirk, M.L. Johnson, T.C. Sander, and D.H. Perrin (2004). Relationship between sex hormones and anterior knee laxity across the menstrual cycle. Med Sci. Sports Exerc.36:1165-1174.
Shultz, S.J., T.C. Sander, S.E. Kirk, and D.H. Perrin (2005). Sex differences in knee joint laxity change across the female menstrual cycle. J. Sports Med Phys Fit.45:594-603.
Urlando, A., and D. Hawkins (2007). Achilles tendon adaptation during strength training in young adults. Med. Sci. Sports Exerc.39:1147-1152.
Vailas, A.C., D.M. Deluna, L.L. Lewis, S.L. Curwin, R.R. Roy, and E.K. Alford (1988). Adaptation of bone and tendon to prolonged hindlimb suspension in rats. J. Appl. Physiol.65:373-376.
West, D.W., A. Lee-Barthel, T. McIntyre, B. Shamim, C.A. Lee, and K. Baar (2015). The exercise-induced biochemical milieu enhances collagen content and tensile strength of engineered ligaments. J. Physiol.593:4665-4675.
Wojtys, E.M., L.J. Huston, T.N. Lindenfeld, T.E. Hewett, and M.L. Greenfield (1998). Association between the menstrual cycle and anterior cruciate ligament injuries in female athletes. Am. J. Sports Med.26:614-619.
Wolfson, R.L., L. Chantranupong, R.A. Saxton, K. Shen, S.M. Scaria, J.R. Cantor,and D. M. Sabatini(2016). Sestrin2 is a leucine sensor for the mTORC1 pathway. Science351:43-48.








