GUT MICROBIOME FUNCTION, STABILITY AND PROFILING, AND RELEVANCE TO ATHLETES
Published
September 2022
Author
Marcus T. O’Brien, Msc, Orla O’Sullivan, PhD, Marcus J. Claesson, PhD, Paul D. Cotter, PhD
Topics

KEY POINTS
- The human gut microbiome is an important and often under-appreciated aspect of human health and may have a significant impact on athletic performance.
- High-level athletes often experience negative health consequences due to high levels of physical activity, many of which may be directly or indirectly linked to the gut microbiome.
- The gut microbiome of high-level athletes undergoes a number of stressors due to their lifestyle, causing high variability and, potentially, long-lasting changes.
- Utilizing microbiome modifying treatments may be of great benefit to elite athletes, especially if used in conjunction with microbiome monitoring.
INTRODUCTION
The human gut microbiome, once an under-appreciated component of human health, over recent years has been the subject of increasingly in-depth investigations. Multiple studies have demonstrated that the gut microbiome has significant and far-reaching effects on gut health, immunity and neurological health (Mohajeri et al., 2018). In recent years, another aspect of the gut microbiome is garnering great attention, its relation to exercise and athlete health as well as performance. This Sports Science Exchange article will discuss how the gut microbiome can affect athletic performance, what the implications are for elite athletes, and suggest potential approaches to monitor and modify the gut microbiome.
THE GUT MICROBIOME
The human gut microbiome, defined as the microbial community inhabiting the human gut, is known to be an important influencer in human health, playing a role in immunomodulation, digestion, and vitamin uptake (Berg et al., 2020; Valdes et al., 2018). The gut microbiome is subject to change, with perturbations in composition and/or function potentially having significant and long-term consequences for health, such as chronic inflammation or the development of conditions such as inflammatory bowel disease (Axelrad et al., 2019; Elias-Oliveira et al., 2020). Under certain circumstances, the gut microbiome may also be positively modified through dietary interventions, exercise or through the use of pro-/pre- or synbiotics, products that contain, or favour the proliferation of, health-promoting organisms (Bengmark, 2001).
It is for these reasons that microbiome profiling has become a quickly growing market, currently valued at over 110 million dollars, and estimated to reach over 800 million dollars by 2030 (InsightAce Analytic Pvt. Ltd, 2022). Many individuals are becoming increasingly interested in tracking their own microbiome development and the implications for their health.
Gut microbiome health is also highly relevant for high-level athletes, who have been noted to have both highly distinct and variable gut microbiome compositions, thought to be caused by the extremes of exercise, diet, and travel they endure (O’Donovan et al., 2020; Xu et al., 2022). For these reasons it is to be expected that there will also be increased interest among high-level athletes and their support teams with respect to the options that are available to monitor and modify their microbiome to ensure optimal health and performance.
THE RELEVANCE OF MICROBIOME HEALTH FOR ATHLETES
The gut microbiomes of athletes and non-athletes have been noted to be significantly different, both in terms of composition and functional capabilities. Athletes’ gut microbiomes are generally more diverse, contain greater levels of health-promoting bacteria (e.g., Akkermansia, Bifidobacterium) and have greater potential to produce certain important bacterial metabolites (Mohr et al., 2020). This implies that physical activity may have a positive effect on gut health. However, this relationship is not as simple as it first seems.
Exercise is believed to have a dose-dependent effect on gut health and immune function, with moderate amounts/intensity of exercise having a positive effect on health, but extreme levels having negative effects, such as increased gastrointestinal distress (Keirns et al., 2020). This is thought to be partially due to intestinal ischemia, the process of redistribution of the blood from the gut to skeletal muscles during intense exercise, increasing gut permeability, allowing bacterial endotoxins to enter the bloodstream, and causing chronic inflammation with knock-on effects for immunity and gastrointestinal health (Moses, 2005).
These negative effects of strenuous exercise can have significant consequences in terms of quality of life and performance for high-level athletes. This is perhaps most pronounced in the extremes of exercise, such as ultra-marathon runners. One study reported that 96% of the participants in a 161 km (100 mile) ultramarathon experienced some form of gastrointestinal symptoms during the race (e.g., belching, nausea, vomiting), with 35.6% of non-finishers attributing their failure to finish the race to these symptoms (Stuempfle and Hoffman, 2015). In addition to this, it is well known that elite athletes suffer from a higher incidence of upper respiratory tract infections (URTI) during training seasons, likely due to the negative effects of extreme exercise on immune function mentioned above (Nieman, 1997). Thankfully, multiple studies have shown that certain probiotics can mitigate both of these negative effects. Schreiber et al. (2021) showed that a multi-strain probiotic (Lactobacillus helveticus Lafti L10, Bifidobacterium animalis ssp. lactis Lafti, Enterococcus faecium R0026, Bifidobacterium longum R0175, and Bacillus subtilis R0179) reduced both gastrointestinal distress and levels of perceived exertion when administered to professional cyclists. Tavares-Silva et al. (2021) showed that a 30-day course of another multi-strain probiotic (Lactobacillus acidophilus LB-G80, Lacticaseibacillus paracasei LPc-G110, Lactococcus subp. lactis LLL-G25, B. animalis subp. lactis BL-G101, and Bifidobacterium bifidum BB-G90) significantly reduced URTI symptoms in marathon runners.
These studies indicate that microorganism-based interventions that act via the gut can have a significant effect on athlete health/performance. In conjunction with this, it is thought that not only can microbiome-based interventions mitigate negative health effects, but they can also improve athletic performance by enhancing muscle growth, endurance, or other functions.
One such microbiome-based function thought to be important in athletic performance is the production of short-chain fatty acids (SCFAs) which are a group of bacterial metabolites thought to be important in human health, largely produced by the bacterial fermentation of dietary fibre. The three most abundant SCFAs produced by the gut microbiome are acetate, propionate, and butyrate, and are thought to provide both additional energy and play a vital role in multiple bodily functions. (Silva et al., 2020). Butyrate is arguably the most important for health in general and has been noted to be anti-inflammatory and modulate important metabolic functions (Amiri et al., 2022). SCFA production, and especially butyrate, has been noted to be increased in the gut microbiomes of athletes (Mohr et al., 2020). Multiple human and animal studies have hypothesised that SCFAs may have a significant effect on athletic performance, aside from their theorized more general positive health effects. One such study noted that peak oxygen uptake in humans, a measure of cardiorespiratory fitness, strongly correlated with the abundance of key butyrate-producing bacteria (Estaki et al., 2016). Furthermore, a study using germ-free mice showed that lean muscle mass could be increased by administering either a gut microbiome sample from healthy mice or a mix of SCFAs (Lustgarten, 2019).
SCFA production has also been noted to be closely linked to bacterial protein metabolism, with many of the undigested proteins we consume being fermented to SCFAs by our gut microbiome. It is thought that bacterial proteolysis not only produces SCFAs, but also increases amino acid availability to the host, potentially further increasing lean muscle gain (Wu et al., 2021). Indeed, studies using a Weizmannia coagulans (previously Bacillus coagulans) probiotic support this hypothesis, by showing increased levels of serum amino acids after a two-week course of the probiotic (Stecker et al., 2020).
A number of studies have noted that gut microbiome composition and microbiome modifying treatments can positively influence endurance. One of the most influential studies in this area noted that Veillonella atypica, a bacterial species known for its lactate metabolism, was enriched in the gut microbiomes of marathon runners post-race (Scheiman et al., 2019). Furthermore, this same study showed that this bacteria could significantly improve run-to-exhaustion time in mice when administered as a probiotic. The authors theorized that the lactate metabolism of Veillonella atypica was improving lactate turnover, decreasing lactic acid build-up in muscles, and thereby improving endurance. While this is an exciting prospect, further study is needed to fully understand this interaction, with some arguing that the choice of control in this paper (Lactobacillus delbrueckii subsp. bulgaricus) may have skewed the results (Fernández-Sanjurjo et al., 2020).
Regardless, when taken together, these studies indicate that a healthy gut microbiome can play a role in an athlete’s performance and, thus, there is the potential to enhance performance through the informed use of microbiome modifying treatments (Figure 1).
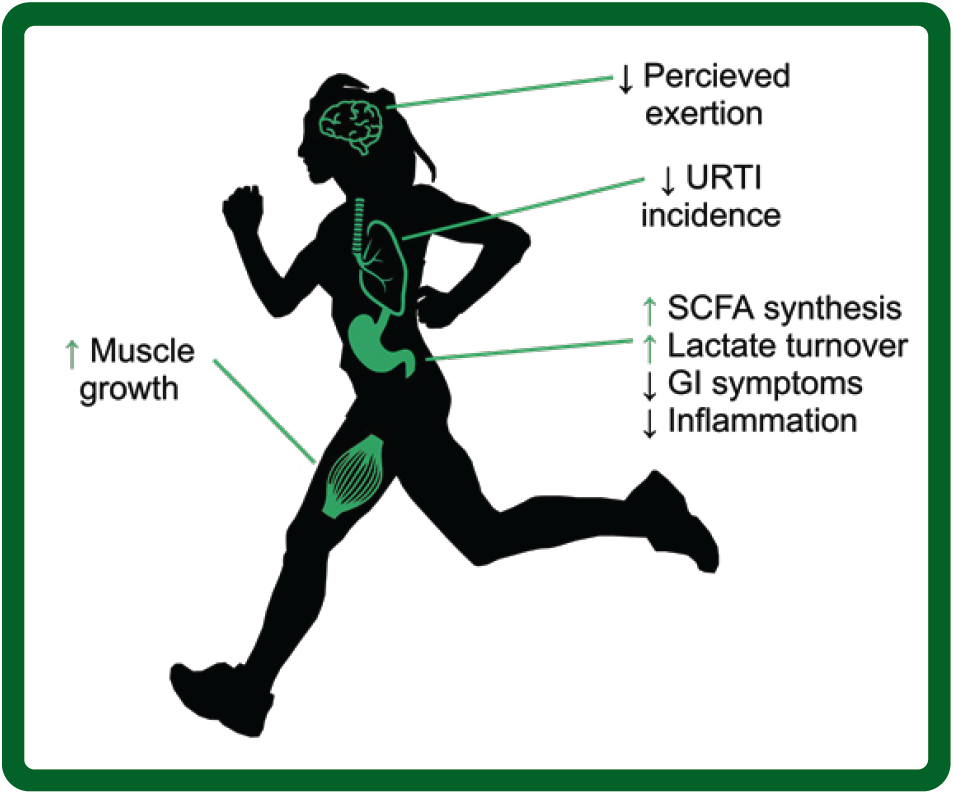 Figure 1: Bodily functions relative to athletes thought to be influenced by the gut microbiome. URTI, upper respiratory tract infections; SCFA, short chain fatty acids; GI, gastrointestinal.
Figure 1: Bodily functions relative to athletes thought to be influenced by the gut microbiome. URTI, upper respiratory tract infections; SCFA, short chain fatty acids; GI, gastrointestinal.MICROBIOME PROFILING
Microbiome profiling is the process of using sequencing technologies to take a “snapshot” of microbiome composition and, in some instances, the functional potential of this microbiome, facilitated by the emergence and rapidly decreasing cost of next-generation sequencing (NGS) technologies (Hamady & Knight, 2009). These technologies allow for the large-scale sequencing and identification of genetic information encoded by the gut microbiome. There are currently multiple companies offering at-home microbiome profiling services, with varying specialties and quality, although without a focus on the relationship between athletics and the microbiome, and in addition to this, few fully utilize longitudinal microbiome profiling, largely focusing on single time point profiling.
While single-point microbiome profiling is useful in certain circumstances, it fails to capture changes in microbial composition, and the implications thereof. While generally stable, the human gut microbiome can change in accordance with diet, age, and disease. These changes are missed when relying on single-point or highly infrequent microbiome profiling (Aleman & Valenzano, 2019). In addition to this, longitudinal microbiome profiling also provides the added benefit of tracking the impacts of pre-/probiotic products, or other modulators including broader dietary changes or the negative impact of antibiotics on the gut microbiome, and also allows for more informed health decisions.
However, it is also vital to note that the field of microbiome profiling is still in its infancy. While multiple correlations between gut microbiome composition and disease states have been noted, there is a need to more fully understand the associated mechanisms involved. This is partially due to the high interindividual variability of the gut microbiome, often making it difficult to tease out significant correlations/patterns (Staley et al., 2018). This arguably increases the importance of longitudinal microbiome monitoring, as while it is difficult to determine significance between two individuals, it is more practical to detect gut microbiome perturbations within a single individual over time.
THE VOLATILITY OF THE ATHLETE GUT MICROBIOME AND THE IMPORTANCE OF GUT MICROBIOME MONITORING
As mentioned above, the gut microbiome is a highly plastic system, which can be sensitive to extreme lifestyle changes. Despite this plasticity, the gut microbiome composition of healthy individuals is generally stable over time, with large perturbations in microbiome composition and diversity generally associated with negative effects or significant lifestyle changes (Fassarella et al., 2021). One study which analysed the microbiome composition of 101 individuals over a one-year time period, noted that microbiome composition and functional capabilities remained relatively stable over time, but certain disease-associated bacteria (e.g. Escherichia coli) were highly variable, even in healthy individuals (Olsson et al., 2022). This supports the hypothesis that longitudinal microbiome monitoring is highly valuable for reliable assessments of microbiome health. This is especially relevant to professional athletes who often subject their microbiomes to the extremes of diet, exercise, and other lifestyle choices.
An athlete's diet and levels of physical activity can change drastically between on-season and off-season (Renard et al., 2021). Diet is a well-known modulator of the gut microbiome and has been observed to cause significant compositional changes within 24 hours of changing diet (Leeming et al., 2019). Based on this, there is potential for extreme and sudden dietary and lifestyle changes to have an impact on an athlete’s gut microbiome. While research in this area is relatively limited, partially due to the lack of longitudinal studies on elite athletes, there is evidence of such a transformation in non-athletes training for a competition. Barton et al. (2021) noted that two individuals who previously described themselves as non-athletes but undertook the challenge to train to complete a marathon and triathlon, respectively, experienced notable shifts in their microbiome diversity in concurrence with training, although in this case, the subjects’ diets remained relatively stable. In addition to this, the travel that professional athletes undertake can have a significant impact on gut microbiome stability and performance. One study from O’Donovan et al. (2020) noted high variability in athletes’ microbiomes associated with travel to specific countries, which in turn was associated with gastrointestinal distress. These studies indicate that the lifestyle choices of professional athletes can have significant effects on the variability of their gut microbiomes, with potential knock-on effects on performance.
Due to the intense stressors athletes often endure, it is a well-known issue that high-level athletes in some sports overuse certain medications, such as anti-inflammatories (Lippi et al., 2006). Frequent use of non-steroidal anti-inflammatory drugs (NSAID) can have a drastic impact on health and the gut microbiome, either by directly affecting the microbiome or by causing gastrointestinal damage, albeit the mechanisms are not yet fully understood (Maseda & Ricciotti, 2020). Multiple studies have shown some promise in being able to mitigate these negative effects through the use of specific probiotics, with some studies showing that specific probiotics could reduce the need for NSAID use in the first place via anti-inflammatory properties (Fu et al., 2021; Maseda & Ricciotti, 2020).
These points indicate that the gut microbiome of elite athletes is more at risk of sudden changes than that of a healthy non-athlete. This emphasises the potential value of consistent microbiome monitoring to high-level athletes. By monitoring the microbiome, any perturbations caused by the above factors could potentially be addressed through the use of microbiome modifying treatments, preventing any potential decrease in performance.
CONCLUSION
Athletes undergo extreme amounts of stress as part of their lifestyle. These stressors, such as changes in diet, travel and extremes of physical activity can have drastic effects on the gut microbiome, which in turn can have knock-on effects on an athlete's performance. For these reasons, the gut microbiome may become an increasingly important aspect to be considered for any athlete. Therefore, close monitoring and careful modification of the gut microbiome may become necessary to achieve optimal performance. However, a substantial amount of research is still required to untangle the relationship between exercise and the gut microbiome before these methods can be fully exploited.
PRACTICAL APPLICATIONS
- Athletes should carefully consider the potential impacts of certain lifestyle choices on their gut microbiome, and the knock-on effects on their performance.
- Certain microbiome modifying treatments may be an avenue for regulating microbiome stability.
- Longitudinal microbiome monitoring may become an important tool in maintaining a healthy athlete microbiome.
The views expressed are those of the authors and do not necessarily reflect the position or policy of PepsiCo, Inc.
REFERENCES
Aleman, F.D.D. and D.R. Valenzano (2019). Microbiome evolution during host aging. PLoS Pathog. 15:e1007727.
Amiri, P., S.A. Hosseini, S, Ghaffari, H. Tutunchi, S. Ghaffari, E. Mosharkesh, S. Asghari, and N. Roshanravan (2021). role of butyrate, a gut microbiota derived metabolite, in cardiovascular diseases: A comprehensive narrative review. Front. Pharmacol. 12:837509.
Axelrad, J.E., O. Olén, J. Askling, B. Lebwohl, H. Khalili, M.C. Sachs, and J.F. Ludvigsson (2019). gastrointestinal infection increases odds of inflammatory bowel disease in a nationwide case-control study. Clin. Gastroenterol. Hepatol. 17:1311–1322.
Barton, W., O. Cronin, I. Garcia-Perez, R. Whiston, E. Holmes, T. Woods, C.B. Molloy, M.G. Molloy, F. Shanahan, P.D. Cotter, and O. O'Sullivan (2021). The effects of sustained fitness improvement on the gut microbiome: A longitudinal, repeated measures case-study approach. Translat. Sports Med. 4:174–192.
Bengmark, S. (2001). Pre-, pro- and synbiotics, Curr. Opin. Clin. Nutr. Metab. Care, 4:571–579.
Berg, G., D. Rybakova, D. Fischer, T. Cernava, M.C. Vergès, T. Charles, X. Chen, L. Cocolin, K. Eversole, G.H. Corral, M. Kazou, L. Kinkel, L. Lange, N. Lima, A. Loy, J.A. Macklin, E. Maguin, T. Mauchline, R. McClure, B. Mitter, M. Ryan, I. Sarand, H. Smidt, B. Schelkle, H. Roume, G.S. Kiran, J. Selvin, R.S.C. Souza, L. van Overbeek, B.K. Singh, M. Wagner, A. Walsh, A. Sessitsch, and M. Schloter (2020). Microbiome definition re-visited: old concepts and new challenges. Microbiome 8:1–22.
Elias-Oliveira, J., J.A. Leite, I.S. Pereira, J.B. Guimarães, G.M.D.C. Manso, J.S. Silva, R.C. Tostes, and D. Carlos (2020). NLR and intestinal dysbiosis-associated inflammatory illness: Drivers or dampers? Front. Immunol. 11:1810.
Estaki, M., J. Pither, P. Baumeister, J.P. Little, S.K. Gill, S. Ghosh, Z. Ahmadi-Vand, K.R. Marsden, and D.L. Gibson (2016). Cardiorespiratory fitness as a predictor of intestinal microbial diversity and distinct metagenomic functions. Microbiome, 4:42.
Fassarella, M., E.E. Blaak, J. Penders, A. Nauta, H. Smidt, and E.G. Zoetendal (2021). Gut microbiome stability and resilience: elucidating the response to perturbations in order to modulate gut health. Gut 70:595–605.
Fernández-Sanjurjo, M., J. Fernández, C. Tomás-Zapico, B. Fernández-García, C.J. Villar, F. Lombó, and E. Iglesias-Gutiérrez (2020). Is physical performance (in mice) increased by Veillonella atypica or decreased by Lactobacillus bulgaricus? J. Sport Health Sci. 9:197–200.
Fu, S.-K., W.C. Tseng, L.W. Tseng, C.C. Lai, Y.C. Tsai, H.L. Tai, and C.C. Hsu (2021). Effect of daily oral PS128 on exercise capacity recovery after a half-marathon. Nutrients 13:4023.
Hamady, M., and R. Knight (2009). Microbial community profiling for human microbiome projects: Tools, techniques, and challenges. Genome Res. 19:1141–1152.
InsightAce Analytic Pvt. Ltd (2022). Human Microbiome-based Gut Health Test and Nutrition Market worth US$ 885.52 Million by 2030 - Exclusive Report by InsightAce Analytic, PR Newswire. Available at: https://www.prnewswire.com/news-releases/human-microbiome-based-gut-health-test-and-nutrition-market-worth-us-885-52-million-by-2030---exclusive-report-by-insightace-analytic-301517714.html (Accessed: 6 April 2022).
Keirns, B.H., N.A. Koemel, C.M. Sciarrillo, K.L. Anderson, and S.R. Emerson (2020). Exercise and intestinal permeability: another form of exercise-induced hormesis? Am. J. Physiol. 319:G512–G518.
Leeming, E.R., A.J. Johnson, T.D. Spector, and C.I. Le Roy (2019). Effect of diet on the gut microbiota: Rethinking intervention duration. Nutrients 11:2862.
Lippi, G., M. Franchini, G.C. Guidi, and W.F. Kean (2006). Non-steroidal anti-inflammatory drugs in athletes. Br. J. Sports Med. 40:661–662; Discussion 662–663.
Lustgarten, M.S. (2019). The Role of the gut microbiome on skeletal muscle mass and physical function: 2019 Update. Front. Physiol. 10:1435.
Maseda, D., and E. Ricciotti (2020). NSAID-gut microbiota interactions. Front. Pharmacol. 11:1153.
Mohajeri, M.H., R.J.M. Brummer, R.A. Rastall, R.K. Weersma, H.J.M. Harmsen, M. Faas, and M. Eggersdorfer (2018). The role of the microbiome for human health: from basic science to clinical applications. Eur. J. Nutr. 57(Suppl 1):1–14.
Mohr, A.E., R. Jäger, K.C. Carpenter, C.M. Kerksick, M. Purpura, J.R. Townsend, N.P. West, K. Black, M. Gleeson, D.B. Pyne, S.D. Wells, S.M. Arent, R.B. Kreider, B.I. Campbell, L. Bannock, J. Scheiman, C.J. Wissent, M. Pane, D.S. Kalman, J.N. Pugh, C.P. Ortega-Santos, J.A. Ter Haar, P.J. Arciero, and J. Antonio (2020). The athletic gut microbiota. J. Int. Soc. Sports Nutr. 17:24.
Moses, F.M. (2005). Exercise-associated intestinal ischemia. Curr. Sports Med. Rep. 4:91–95.
Nieman, D.C. (1997). Risk of upper respiratory tract infection in athletes: an epidemiologic and immunologic perspective. J. Athl. Train. 32:344–349.
O’ Donovan, C.M., B. Connor, S.M. Madigan, P.D. Cotter, and O. O’Sullivan (2020). Instances of altered gut microbiomes among Irish cricketers over periods of travel in the lead up to the 2016 World Cup: A sequencing analysis. Travel Med. Infect. Dis. 35:101553.
Olsson, L.M., F. Boulund, S. Nilsson, M.T. Khan, A. Gummesson, L. Fagerberg, L. Engstrand, R. Perkins, M. Uhlén, G. Bergström, V. Tremaroli, and F. Bäckhed (2022). Dynamics of the normal gut microbiota: A longitudinal one-year population study in Sweden. Cell Host Microbe 30:726-739.
Renaud, M., D.T. Kelly, N.N. Chéilleachair, and C.O. Catháin (2021). How does the dietary intake of female field-based team sport athletes compare to dietary recommendations for health and performance? A systematic literature review. Nutrients 13:1235.
Scheiman, J., J.M. Luber, T.A. Chavkin, T. MacDonald, A. Tung, L.D. Pham, M.C. Wibowo, R.C. Wurth, S. Punthambaker, B.T. Tierney, Z. Yang, H.W. Hattab, J. Avila-Pacheco, C.B. Clish, S. Lessard, G.M. Church, and A.D. Kostic (2019). Meta-omics analysis of elite athletes identifies a performance-enhancing microbe that functions via lactate metabolism. Nature Med. 25:1104–1109.
Schreiber, C., S. Tamir, R. Golan, A. Weinstein, and Y. Weinstein (2021). The effect of probiotic supplementation on performance, inflammatory markers and gastro-intestinal symptoms in elite road cyclists. J. Int. Soc. Sports Nutr. 18:36.
Silva, Y.P., A. Bernardi, and R.L. Frozza (2020). The role of short-chain fatty acids from gut microbiota in gut-brain communication. Front. Endocrinol. 11:25.
Staley, C., T. Kaiser, and A. Khoruts (2018) ‘Clinician guide to microbiome testing. Digest. Dis. Sci. 63:3167–3177.
Stecker, R.A., J.M. Moon, T.J. Russo, K.M. Ratliff, P.W. Mumford, R. Jäger, M. Purpura, and C.M. Kerksick (2020). GBI-30, 6086 improves amino acid absorption from milk protein. Nutr. Metab. 17:93.
Stuempfle, K.J., and M.D. Hoffman (2015). Gastrointestinal distress is common during a 161-km ultramarathon. J. Sports Sci. 33:1814–1821.
Tavares-Silva, E., A.V. Caris, S.A. Santos, G.R. Ravacci, and R.V. Thomatieli-Santos (2021) ‘Effect of multi-strain probiotic supplementation on URTI symptoms and cytokine production by monocytes after a marathon race: A randomized, double-blind, placebo study. Nutrients 13:1478.
Valdes, A.M., J. Walter, E. Segal, and T.D. Spector (2018). Role of the gut microbiota in nutrition and health. Br. Med. J. 361:k2179.
Wu, L., Z. Tang, H. Chen, Z. Ren, Q. Ding, K. Liang, and Z. Sun (2021). Mutual interaction between gut microbiota and protein/amino acid metabolism for host mucosal immunity and health. Anim. Nutr. 7:11–16.
Xu, Y., F. Zhong, X. Zheng, H.-Y. Lai, C. Wu, and C. Huang (2022). Disparity of gut microbiota composition among elite athletes and young adults with different physical activity independent of dietary status: A matching study. Front. Nutr. 9:843076.








