KEY POINTS
- Free radicals (radicals) are molecules with an unpaired electron in their outer orbital. The radicals are a highly reactive chemical species capable of damaging muscle fiber components such as proteins and lipids.
- Muscular contractions (i.e., exercise) promote the production of radicals in the active skeletal muscles. The magnitude of radical production in the working muscles increases as a function of both exercise intensity and duration.
- Intense and prolonged endurance exercise can damage muscle components; this is referred to as exercise-induced oxidative stress.
- To protect against radical-mediated damage, muscle fibers and other cells synthesize a variety of molecules that scavenge radicals and prevent oxidative damage. Collectively, these molecules are called endogenous antioxidants.
- Exogenous antioxidants (i.e., antioxidants obtained in the diet) interact with endogenous antioxidants to form a cooperative network of protection against radical-mediated cellular damage.
INTRODUCTION
Free radicals (hereafter referred to as radicals) are molecules or fragments of molecules that contain an unpaired electron in their outer orbital (Powers & Jackson, 2008). This unpaired electron means that radicals are highly reactive and high levels of radical production in cells result in oxidative damage to key cellular components; this radical-mediated cellular damage is referred to as oxidative stress.
It is well established that contracting skeletal muscles produce radicals and the rate of radical production in muscles rises as the exercise intensity is increased (Powers & Jackson, 2008). Given that radical production in muscle fibers can damage cellular components, it is not surprising that muscle fibers contain protective molecules called antioxidants that defend against radical-mediated damage. Two major classes of antioxidants exist: 1) endogenous antioxidants and 2) exogenous antioxidants. Endogenous antioxidants are proteins and polypeptides that are synthesized in the cell while exogenous antioxidants are obtained from the ingestion of fruits and vegetables. Additionally, antioxidants can be attained via commercially available dietary supplements.
Exercise induces radical production and the resultant exercise-induced oxidative stress in skeletal muscles impairs muscle force production and contributes to muscle fatigue during prolonged endurance exercise (Reid, 2008). The knowledge that exercise-induced radical production can damage skeletal muscle fibers and contribute to fatigue has motivated many endurance athletes to consume antioxidant supplements.
This Sports Science Exchange article is the first of a two-part series that discusses exercise, oxidative stress and antioxidant supplementation. The objective of “part one” of this exposé is to provide an overview of exercise-induced oxidative stress and to introduce endogenous (cellular antioxidants) and exogenous antioxidants (i.e., dietary antioxidants). This primer will set the stage for “part two” of this article which will tackle two important issues: 1) Should athletes supplement with antioxidants to protect against exercise-induced oxidative stress?; and 2) Does antioxidant supplementation improve endurance exercise performance? The current article begins with a discussion of exercise-induced radical production and oxidative stress.
Exercise-induced radical production
The first evidence that contracting muscles produce radicals was reported over 30 years ago (Davies et al., 1982) and it is now well established that muscular contractions (i.e., exercise) induce radical production (Powers & Jackson, 2008). Emerging evidence indicates that contraction-induced radical production occurs primarily in the cytosol of the muscle and the magnitude of exercise-induced radical production is influenced by several factors, including environmental conditions and the intensity and duration of exercise (Powers et al., 2011; Sakellariou et al., 2013). Specifically, skeletal muscle radical production increases as a function of both the exercise intensity and duration. Moreover, contracting skeletal muscles produce more radicals during exercise in a hot environment and during work at high altitude (i.e., ~4,000 meters) (Arbogast & Reid, 2004; Radak et al., 1994). Therefore, the magnitude of exercise-induced muscle radical production can range from low to high levels of oxidants depending upon the exercise conditions.
Although contracting skeletal muscles produce radicals, exercise bouts do not always result in oxidative damage to skeletal muscles. For example, low-intensity and short-duration exercise does not generally promote oxidative stress in skeletal muscles (Powers & Jackson, 2008). Nonetheless, prolonged endurance exercise performed at moderate-to-high intensities often results in oxidative damage to skeletal muscles of untrained individuals. Note, however, that highly-trained endurance athletes have well-adapted endogenous antioxidant buffer systems in their skeletal muscles that resist exercise-induced oxidative stress (Powers & Jackson, 2008). Therefore, whether an exercise bout results in oxidative stress is dependent upon several factors, including the intensity and duration of exercise as well as the exercise training status of the individual.
Overview of antioxidants
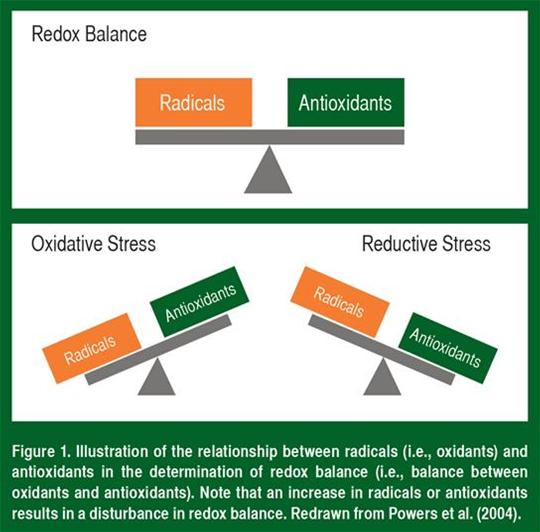
The balance between antioxidants and oxidants is commonly referred to as “redox balance” and is illustrated in Figure 1. Oxidative stress results from an imbalance between antioxidants and oxidants (i.e., radicals); this occurs when oxidant production exceeds the antioxidant capacity (Figure 1). In contrast, reductive stress occurs when the antioxidant capacity greatly exceeds the rate of oxidant (i.e., radical production).
Muscle fibers are protected against oxidative stress by a complex system of endogenous and exogenous antioxidants. More specifically, a coordinated network of enzymatic and non-enzymatic antioxidants exists in both the intracellular and extracellular locations to remove radicals before they damage proteins, lipids or DNA. Enzymatic antioxidants are cellular proteins that catalytically remove radicals or reactive species to protect against oxidative stress. Non-enzymatic antioxidants are molecules (e.g., glutathione or antioxidants contained in food) that can eliminate radicals by means other than a catalytic reaction. To provide optimal protection against radical-mediated damage, both enzymatic and non-enzymatic antioxidants are strategically compartmentalized (e.g., organelles vs. membrane vs. cytosol) throughout the cell (Powers & Jackson, 2008).
Several strategies are applied by both endogenous and exogenous antioxidants to protect against oxidant-mediated injury. These include converting radicals into non-radicals (i.e., scavenging) and preventing the conversion of relatively inactive radicals into more damaging species (Powers et al., 2011). A brief overview of both endogenous and exogenous antioxidants follows.
Endogenous antioxidants
Endogenous antioxidants are synthesized in cells and include both enzymatic and non-enzymatic antioxidants. Key enzymatic antioxidants include superoxide dismutase (SOD), glutathione peroxidase (GPX) and catalase (CAT). Together, these antioxidant enzymes prevent oxidative stress by scavenging radicals and reactive species before they damage cellular components. The major non-enzymatic antioxidant in all cells is glutathione (GSH). This important non-enzymatic antioxidant can act not only as an independent oxidant scavenger but it can also work with glutathione peroxidase to remove hydrogen peroxide (an oxidant) from the cell. Collectively, enzymatic and non-enzymatic antioxidants work as a unit to protect cells against radical-mediated oxidative injury. It is important to note that endurance exercise training increases the expression of enzymatic antioxidants in skeletal muscle to provide protection against exercise-induced oxidative stress (Powers & Jackson, 2008). Therefore, compared to untrained individuals, well-trained endurance athletes possess higher levels of endogenous antioxidants in their trained skeletal muscles.
Exogenous antioxidants
Most fruits and vegetables contain a variety of antioxidants and together with endogenous antioxidants, exogenous antioxidants consumed in the diet also contribute to cellular protection against radicals. Important dietary antioxidants include vitamin E (tocopherols and tocotrienols), vitamin C (ascorbic acid), carotenoids (e.g., β-carotene), flavonoids, alpha lipoic acid and several trace minerals. A brief introduction to each of these different classes of antioxidants follows.
The generic term vitamin E refers to at least eight structural isomers of tocopherols or tocotrienols (Traber & Stevens, 2011). Among these, α-tocopherol is the best known and possesses the most antioxidant activity (Janero, 1991). Vitamin E is lipid soluble and remains one of the most widely distributed antioxidants in nature and protects cell membranes against radical-mediated damage (Traber & Stevens, 2011). Although vitamin E is an efficient radical scavenger, the interaction of vitamin E with a radical results in a decrease in function of vitamin E and the formation of the vitamin E radical. However, this vitamin E radical can be recycled back to its native state by other antioxidants (e.g., vitamin C). Under most dietary conditions, the concentration of vitamin E in tissues is relatively low and vitamin E deficiency has been shown to promote muscle fatigue during exercise (Coombes et al., 2002). However, vitamin E levels in the tissues can be elevated with dietary supplementation. Indeed, the fact that vitamin E is lipid soluble means that supplementation with megadoses of vitamin E can result in large increases in body stores of this vitamin. Too much vitamin E in the body, however, can result in a state of vitamin E toxicity with gastric distress and an increased risk of bleeding due to the anticoagulant properties of vitamin E (Herbert, 1994).
Carotenoids are natural pigments synthesized in many plants and are responsible for the bright colors of many fruits and vegetables (Paiva & Russell, 1999). Over 600 carotenoids exist in nature and most of these carotenoids have antioxidant activity (e.g., β-carotene). Similar to vitamin E, carotenoids are lipid soluble antioxidants and because of their location in cell membranes and their radical scavenging capacity, carotenoids are efficient biological antioxidants against radical-mediated damage to cell membranes (Krinsky, 1998).
In contrast to both vitamin E and the carotenoids, vitamin C (ascorbic acid) is hydrophilic and therefore, is located in the aqueous compartment (i.e., cytosol) of the cell. As an antioxidant, vitamin C performs two key functions. First, vitamin C can directly scavenge numerous radical species (Carr & Frei, 1999) and secondly, vitamin C plays an important role in the recycling of vitamin E. Therefore, vitamin C and E work together to protect the cell against radical-mediated damage (Traber & Stevens, 2011). Similar to vitamin E, some authors argue that megadoses of vitamin C may also have negative health consequences (Herbert, 1994).
Flavonoids are a large family of compounds (>4,000 members) found in many plants. At present, the antioxidant properties of many naturally occurring flavonoids have not been investigated. Nonetheless, numerous flavonoids have been studied (e.g., catechins, quercetin, etc.) and have been shown to possess a variety of important biological activities including both anti-inflammatory and antioxidant properties (Myburgh, 2014). Although many flavonoids possess antioxidant properties, the radical scavenging activity of flavonoids varies widely among family members.
Alpha lipoic acid (α-LA) is a naturally occurring compound that can be obtained from a variety of foods including spinach, broccoli, tomatoes, peas and Brussels sprouts (Shay et al., 2009). Further, humans can synthesize α-LA in very small quantities. Normally, α-LA is found in small quantities in body tissues and is bound to an enzyme complex that limits its function as an antioxidant. However, unbound, α-LA and several of its metabolites are effective antioxidants. Further, α-LA can provide additional antioxidant effects by recycling both vitamin C and vitamin E (Coombes et al., 2000).
It is also noteworthy that several trace minerals play important but indirect roles in providing antioxidant protection in cells. Trace minerals involved in antioxidant-related functions include copper, zinc, iron, selenium and manganese. These minerals contribute to the body’s antioxidant defenses by acting as co-factors for antioxidant enzymes.
To summarize, many different types of antioxidants are contained in fruits and vegetables. Because different classes of dietary antioxidants vary in their radical scavenging capacity and are positioned in different cellular locales (e.g., membrane vs. cytosol vs. organelles), it is important to consume a wide variety of these antioxidants to ensure that a thorough balance of exogenous antioxidants are present in cells. The important interplay between exogenous and endogenous antioxidants will be discussed in the next section.
Interactions between endogenous and exogenous antioxidants
The previous sections have provided a “big picture” overview of both endogenous and exogenous antioxidants. However, it is important to appreciate that endogenous and exogenous antioxidants work as a team to optimize protection against radical-mediated cellular damage. Indeed, as discussed previously, some dietary antioxidants are located in the lipid-based cell membranes (e.g., vitamin E and carotenoids), whereas other dietary antioxidants are found in the aqueous phase (i.e., cytosol) of the cell (e.g., vitamin C) (Figure 2). Collectively, these dietary (exogenous) antioxidants assist the endogenous antioxidant system by scavenging radicals that were not removed by the endogenous antioxidant system. Moreover, no endogenous antioxidants exist in cell membranes. Therefore, the exogenous (dietary) antioxidants vitamin E and carotenoids are essential to protect cell membranes and the membranes surrounding organelles (e.g., mitochondria) from radical-mediated damage.

As mentioned previously, several dietary antioxidants synergistically interact to replenish specific exogenous antioxidants. Indeed, α-LA is capable of recycling vitamin C. Moreover, vitamin C can replenish vitamin E by recycling the vitamin E radical back to the active antioxidant form of vitamin E. This recycling process is an important and valuable tool to ensure optimal antioxidant protection in the cell.
Finally, because of the varied locations and antioxidant capacities of different antioxidants, it is clear that consumption of a single antioxidant in the diet (e.g., vitamin C) is not sufficient to provide optimal protection from radical-mediated damage in cells. Therefore, this fact provides the foundation for the recommendation that athletes consume a varied diet that is rich in fruits and vegetables (Powers et al., 2004). Further, in addition to the well-known antioxidant vitamins contained in foods, fruits and vegetables contain a variety of phytochemicals with antioxidant properties that interact with vitamins to provide protection against oxidative stress.
PRACTICAL IMPLICATIONS
- Regular bouts of endurance exercise results in increased endogenous antioxidant enzymes in the trained skeletal muscles. This translates to an improved ability to protect against exercise-induced oxidative stress in skeletal muscles.
- Many fruits and vegetables contain numerous compounds with antioxidant activity.
- Because dietary antioxidants differ in their cellular locations and radical scavenging capacity, it appears prudent to consume a wide variety of fruits and vegetables to optimize intake of nutritional antioxidants.
- Antioxidants exist in small quantities in foods and therefore, there is limited risk of an antioxidant “overdose” by consuming a diet rich in both fruits and vegetables. However, the ingestion of megadoses of antioxidant via dietary supplements (e.g., vitamin E) can increase the risk of toxicity and the associated possibility of negative health consequences.
SUMMARY
Exercise promotes radical production in the working muscles and prolonged/intense exercise can produce an imbalance between radical production and muscle antioxidants altering the “redox balance” and resulting in oxidative stress. To protect against radical-mediated damage, muscle cells contain endogenous antioxidants to scavenge radicals. Moreover, exogenous antioxidants obtained in the diet cooperate with endogenous antioxidants to form a supportive network of cellular protection against radical-mediated oxidative stress. In regard to exogenous antioxidants, a varied diet of fruits and vegetables is a sensible means of obtaining a balance of exogenous antioxidants. In contrast, because of the risk of negative consequences, consuming megadoses of antioxidants via supplements is not recommended.
REFERENCES
Arbogast, S., and M.B. Reid (2004). Oxidant activity in skeletal muscle fibers is influenced by temperature, CO2 level, and muscle-derived nitric oxide. Am. J. Physiol. 287:R698-R705.
Carr, A., and B. Frei (1999). Does vitamin C act as a pro-oxidant under physiological conditions? FASEB J. 13:1007-1024.
Coombes, J.S., S.K. Powers, H.A. Demirel, J. Jessup, H.K. Vincent, K.L. Hamilton, H. Naito, R.A. Shanely, C.K. Sen, L. Packer, and L.L. Ji (2000). Effect of combined supplementation with vitamin E and alpha-lipoic acid on myocardial performance during in vivo ischaemia-reperfusion. Acta Physiol. Scand. 169:261-269.
Coombes, J.S., B. Rowell, S.L. Dodd, H.A. Demirel, H. Naito, R.A. Shanely, and S.K. Powers (2002). Effects of vitamin E deficiency on fatigue and muscle contractile properties. Eur. J. Appl. Physiol. 87:272-277.
Davies, K.J., A.T. Quintanilha, G.A. Brooks, and L. Packer (1982). Free radicals and tissue damage produced by exercise. Biochem. Biophys. Res. Commun. 107:1198-1205.
Herbert, V. (1994). The antioxidant supplement myth. Am. J. Clin. Nutr. 60:157-158.
Janero, D.R. (1991). Therapeutic potential of vitamin E against myocardial ischemic-reperfusion injury. Free Radic. Biol. Med. 10:315-324.
Krinsky, N.I. (1998). The antioxidant and biological properties of the carotenoids. Ann. NY Acad. Sci. 854:443-447.
Myburgh, K.H. (2014). Polyphenol supplementation: benefits for exercise and oxidative stress? Sports Med. 44:S57-S70.
Paiva, S.A., and R.M. Russell (1999). Beta-carotene and other carotenoids as antioxidants. J. Am. Coll. Nutr. 18:426-433.
Powers, S.K., K.C. DeRuisseau, J. Quindry, and K.L. Hamilton (2004). Dietary antioxidants and exercise. J. Sports Sci. 22:81-94.
Powers, S.K., and M.J. Jackson (2008). Exercise-induced oxidative stress: cellular mechanisms and impact on muscle force production. Physiol. Rev. 88:1243-1276.
Powers, S.K., L.L. Ji, A.N. Kavazis, and M.J. Jackson (2011). Reactive oxygen species: impact on skeletal muscle. Compr. Physiol. 1:941-969.
Radak, Z., K. Lee, W. Choi, S. Sunoo, T. Kizaki, S. Oh-ishi, K. Suzuki, N. Taniguchi, H. Ohno, and K. Asano (1994). Oxidative stress induced by intermittent exposure at a simulated altitude of 4000 m decreases mitochondrial superoxide dismutase content in soleus muscle of rats. Eur. J. Appl. Physiol. Occup. Physiol. 69:392-395.
Reid, M.B. (2008). Free radicals and muscle fatigue: Of ROS, canaries, and the IOC. Free Radic. Biol. Med. 44:169-179.
Sakellariou, G.K., A. Vasilaki, J. Palomero, A. Kayani, L. Zibrik, A. McArdle, and M.J. Jackson (2013). Studies of mitochondrial and nonmitochondrial sources implicate nicotinamide adenine dinucleotide phosphate oxidase(s) in the increased skeletal muscle superoxide generation that occurs during contractile activity. Antioxid. Redox Signal. 18:603-621.
Shay, K. P., R. Moreau, E. J. Smith, A. R. Smith, and T. M Hagen. (2009) Alpha lipoic acid as a dietary supplement: Molecular mechanisms and therapeutic potential. Biochim. Biophys. Acta 1790:1149–1160
Traber, M.G., and J.F. Stevens. (2011). Vitamins C and E: beneficial effects from a mechanistic perspective. Free Radic. Biol. Med. 51:1000-1013.