KEY POINTS
-
American football is a high-intensity intermittent sport that results in muscle, cartilage and bone damage that must be minimized so that players are healthy at the end of the year.
-
The key to optimal muscle function is to maximize protein turnover while maintaining positive protein balance.
-
The regeneration of damaged muscle is dependent on a protein kinase complex called the mechanistic/mammalian target of rapamycin complex 1 (mTORC1).
-
mTORC1 can be activated by a diet rich in proteins that are rapidly absorbable and rich in the amino acid leucine. mTORC1 can also be inactivated by the consumption of alcohol.
-
The intake of Omega-3 fatty acids can improve muscle protein synthesis, decrease inflammation and improve cognitive function.
-
Over 50% of athletes in a recent meta-analysis had insufficient levels of circulating vitamin D (<32 ng/mL) regardless of ethnicity. This is important because adequate vitamin D status improves muscle, bone and immune function.
-
Athletes with darker skin tones are at greater risk of vitamin D insufficiency/deficiency because they do not synthesize vitamin D from the sun as efficiently.
-
A simple nutritional strategy is presented that can be used to maximize recovery during an American football season.
FUNCTIONAL DEMANDS OF AMERICAN FOOTBALL
In addition to the metabolic demands of American football, detailed in SSE 143, the physical toll can be extreme. One measure of the physical toll is creatine kinase (CK) release into the blood as a result of muscle damage (Zimmerman & Shen, 2013). Blood levels of CK following football games rise progressively during the season (Kraemer et al., 2013), indicating a cumulative toll of the football season on muscle health. This must be minimized so that players can perform their best in the games that matter most at the end of the season. On top of the muscular damage, the impact of the game on cartilage (Gray et al., 2013) and bone (Mall et al., 2012) health also needs to be addressed during recovery.
As with all elite sport, the primary approaches to protect an athlete from injury and maximize performance are to optimize training and recovery. This SSE will focus on dietary principles to optimize recovery with a primary focus on foods that can help an athlete recover and prepare for games during a taxing football season.
METABOLIC RECOVERY USING DIET
American football will preferentially deplete glycogen and this can drive dehydration. Therefore, in the hours and days after a game, and in the days before the next game, the focus is first and foremost on the replenishment and recovery of glycogen and fluids. The recovery of glycogen, and adequate kilocaloric intake, macronutrient breakdown, nutrient timing and general dietary needs of football players in season as well as how to fuel maximal athletic performance is detailed in SSE #146; while optimizing fluid balance will be discussed in SSE #141. This SSE will therefore focus on nutrients that are specifically needed to maintain muscle, bone, cartilage, immune and brain health during a football season.
MUSCLE RECOVERY — PROTEIN TO MAXIMIZE TURNOVER
The key to long-term muscle health is the ability to rapidly increase protein turnover in response to trauma and maintain protein balance. Increasing protein turnover means increasing the rate of both protein synthesis and breakdown within muscle. When this happens, the result is better muscle function (Hwee et al., 2014). Increasing protein turnover is essential during the football season in order to repair muscle fibers damaged during training or games (Kraemer et al., 2013). Proteins are broken down in order to replace damaged fibers and provide a stimulus for muscle repair. As a result, protein breakdown is proportional to protein synthesis (Phillips et al., 1997), and trained athletes can recycle the amino acids that are released from the breakdown of damaged muscle (Phillips et al., 1999), resulting in larger and stronger muscles when turnover is high (Hwee et al., 2014).
Protein balance on the other hand is the arithmetic sum of the rate of protein synthesis minus protein degradation. In order for a muscle to stay the same size, protein balance must be zero, for it to grow balance must be positive, and for it to get smaller muscle protein balance must be negative. To maintain strength, speed and agility throughout a physically demanding football season, every football player wants to maintain protein balance as close to zero as possible in season.
Football can be thought of as a series of high load lengthening contractions with an eccentric bias. What this means is that playing football is like a very heavy resistance exercise bout that uses a lot of eccentric or plyometric movements. In other words, this is an injury stimulus that can lead to larger, stronger muscles if given optimal recovery time. We know that after resistance exercise both muscle protein synthesis and degradation increase (Phillips et al., 1997). If the exercise is performed in the fasted state, the increase in protein degradation is greater than the increase in synthesis resulting in net muscle breakdown (Tipton et al., 1999). In order for muscle protein balance to return to zero or become positive, the athlete has to consume protein, especially proteins rich in essential amino acids (Tipton et al., 1999).
Central to both the regeneration of muscle following injury and the increase in protein synthesis after exercise is a protein complex called the molecular/mammalian target of rapamycin complex 1 (mTORC1). Following resistance exercise, the rate of muscle growth is directly proportional to the activity of mTORC1 (Baar & Esser, 1999) and mTORC1 activity is necessary for either resistance exercise (Drummond et al., 2009) or amino acids (Dickinson et al., 2011) to increase protein synthesis. Further, mTORC1 is also required for muscle to regenerate after injury (Ge et al., 2009). Therefore, in order for muscle to regenerate and grow stronger after it has been damaged during a football game, mTORC1 needs to be activated.
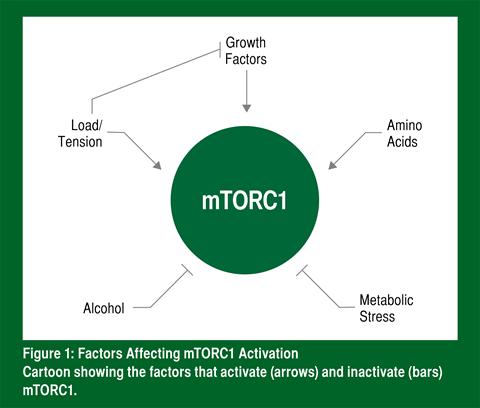
In the roughest sense, mTORC1 can be activated by three things: 1) exercise; 2) hormones or growth factors; and 3) amino acids (Figure 1). When heavy exercise is performed, it blocks the ability of growth factors to activate mTORC1, but activates mTORC1 directly (Hamilton et al., 2014). Amino acids (from the diet) and heavy exercise (games, practices, or strength and conditioning) activate mTORC1 in different ways and therefore the effects are additive. This means for a football player in season there are functionally two ways to activate mTORC1. More specifically, mTORC1 is activated by high loads to failure (Baar & Esser, 1999) and the amino acid leucine (Moberg et al., 2014). Even though all of the other amino acids are needed to increase protein synthesis (Churchward-Venne et al., 2012), leucine is the trigger that turns on mTORC1 and drives a positive protein balance (Breen & Churchward-Venne, 2012). Therefore, to turn on mTORC1 and maximize muscle regeneration and protein balance, athletes should consume a diet founded on leucine-rich proteins.
On the other side of the equation, two things are known to turn mTORC1 off: 1) alcohol; and 2) metabolic stress. A blood alcohol level of 0.06 g/100 mL is enough to decrease muscle protein synthesis after exercise, likely the result of lower mTORC1 activity (Parr et al., 2014). Because mTORC1 activation is needed for proper muscle repair, if alcohol is consumed after a game or a hard training session, it is likely that the athlete will not recover properly or recovery will be significantly delayed. Much like alcohol, metabolic stress due either to high-intensity interval training (Coffey et al., 2009) or an energy deficit (Pasiakos et al., 2014) can decrease mTORC1 activity and muscle protein synthesis. Therefore, during the season, athletes should strive to maintain energy balance, and strength training should never be performed either immediately before or after practice.
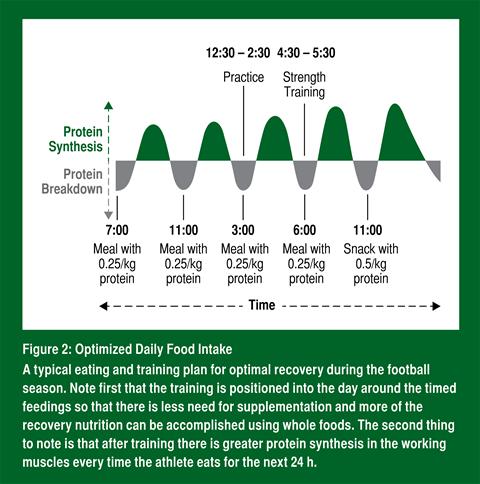
Beyond the basic ideas as to how leucine-rich foods and mTORC1 are good for optimizing protein balance and recovery during a football season, the precise quantity and timing of protein intake is known as well. In order to maximize protein synthesis, an athlete should consume 0.25 g/kg body mass of leucine-rich protein immediately after training (Moore et al., 2009) and every ~4 h throughout the day. This amount is selected because taking in more protein does not result in any further increase in muscle protein synthesis. This also means taking more protein less often cannot compensate for regularly spaced meals each containing the optimal amount of protein (Areta et al., 2013); (Figure 2).
OMEGA 3 FATTY ACIDS AND RECOVERY
An Omega-3 fatty acid is a polyunsaturated fatty acid, meaning it contains two or more double bonds with one of the double bonds at the third carbon from the methyl end. These polyunsaturated fatty acids are considered essential because the human body cannot manufacture them in appreciable amounts.
Two long chain Omega-3 fatty acids will be discussed in detail: eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA). Other Omega-3 fatty acids, like α/γ-linolenic acid (ALA), can be converted to EPA and DHA within the body. However, the conversion rate of ALA to EPA and DHA in young men is approximately 8% and 0-4%, respectively (Burdge et al., 2002).
The Omega-3 fats are the focus of a great deal of performance research since they are known to incorporate into the membranes of cells, improve muscle protein synthesis, decrease inflammation and improve cognitive function. All of these have performance and health implications for athletes.
Muscle Protein Synthesis
The majority of studies examining the impact of Omega-3 fatty acids on muscle protein synthesis have been conducted in older populations (>60 yr) or animals. In one study, Omega-3 fatty acid intake resulted in greater activation of mTORC1 during periods of high insulin and amino acid infusions in older adults (Smith et al., 2011a). In a follow-up study, the authors found that the anabolic response to insulin and amino acid infusion was greater after supplementation with 4 g of long chain Omega-3s (including 1.86 g EPA, 1.50 g DHA) daily for 8 wk (Smith et al., 2011b). These data support the hypothesis that when insulin and amino acid levels are high, such as following a large meal, adequate amounts of Omega-3 fatty acids may improve muscle protein synthesis through the activation of mTORC1. While these data are exciting, more research is necessary to establish whether these findings extend to elite athletes.
Inflammation and Muscle Soreness
American football results in significant amounts of damage to muscle and this damage accumulates as the season progresses. One of the natural responses to damage is an inflammatory response within the muscle that is necessary for training adaptations to occur (Koh & Pizza, 2009). Too much inflammation, however, can impair the ability of the muscle to recover post-exercise. Omega-3 fatty acids have natural anti-inflammatory properties. DHA and EPA decrease the expression of inflammatory cytokines and also give rise to a family of anti-inflammatory mediators, termed resolvins (Calder, 2006). DHA also specifically influences the activity of inflammatory cells, altering neutrophil proliferation and monocyte phagocytosis (Gogus, 2010).
One result of the anti-inflammatory effects of EPA and DHA is to decrease muscle soreness. Two studies have shown a direct impact of Omega-3 intake on delayed onset muscle soreness (DOMS) 48 h post-exercise. Tartibian et al. (2009) had untrained subjects consume 1.8 g/d of an Omega-3 supplement (including 0.324 g EPA, 0.216 g DHA) for 30 d before completing an eccentric exercise session. Subjects who took the Omega-3 supplement reported reductions in perceived pain and displayed improved knee range of motion 48 h post-exercise, even at this low dose. Jouris and colleagues (2011) similarly showed a decrease in DOMS, as a result of Omega-3 supplementation (including 2 g EPA, 1 g DHA/d) 48 h after an eccentric exercise protocol. These exciting results suggest that even low amounts of Omega-3 fatty acids can decrease DOMS. Additional research in highly trained individuals is needed, but there appears to be potential for Omega-3 intake to improve muscle recovery.
Cognitive Function
DHA plays a very specific and essential role in the functioning of neural tissue (Dyall & Michael-Titus, 2008). When examining the structure of the phospholipid bilayer of neurons, DHA is the most prevalent Omega-3 fatty acid and is also involved in the synthesis of neuroprotectins (antioxidant, antiapoptotic and anti-inflammatory molecules). The prevalence of DHA within neural tissues impacts cognitive performance, memory and learning ability. These factors all have implications for team-based sports such as American football. For example, when Fontani and colleagues (2005) supplemented young adult subjects with 4 g/d of Omega-3 (including 1.6 g EPA, 0.8 g DHA) for 35 d they found that subjects performed better on cognitive tests, had faster reaction times, as well as a prolonged attention span. This is one reason why Omega-3 (particularly DHA) supplementation now plays a significant role in traumatic brain injury (concussion) rehabilitation protocols.
Sources and Recommendations for Omega-3 Supplementation
ALA is an Omega-3 derived from plant base sources, such as flax seeds, flaxseed oil, nuts, nut butters, algae, seeds, soybean oil and rapeseed oil. EPA and DHA are Omega-3 fatty acids found within animal sources such as cold water fatty fish (e.g., tuna, salmon), fish oils and krill oil. Many varieties of Omega-3 supplements also exist. It is important for an athlete to note that there is a difference between “grams of fish oil” and “grams of EPA/DHA” on a supplement label.
The United States Institute of Medicine recommends that men consume 1.6 g per day of ALA. There are no established recom-mendations for EPA and DHA intake, although the American Heart Association has provided guidelines for EPA and DHA intake (Kris-Etherton et al., 2002) (Table 1).

Given the importance of Omega-3 fatty acids in recovery, immunity and brain function, athletes without bleeding issues should consume 3 g/d of Omega-3 fatty acids with ~0.75 g EPA and 1 g DHA and the remainder from plant sources.
VITAMIN D
Serum Vitamin D Levels
In a recent systematic-review and meta-analysis of studies looking at vitamin D status in athletes, over half (56%) of athletes had inadequate levels, defined as <32 ng/mL (Farrokhyar et al., 2015). Specifically in the National Football League, of the 80 players tested in one study, 55 had either insufficient (12-20 ng/mL) or deficient (<12 ng/mL) levels of serum vitamin D (Maroon et al., 2015). The implications of insufficient or deficiency of serum vitamin D on bone health, immunity (Schwalfenberg, 2011), neuromuscular function, cell growth and inflammation are significant (Ross et al., 2011). Therefore, using blood tests throughout the season to identify potential problems before they develop is essential.
Musculoskeletal Health and Recovery
The role of vitamin D in musculoskeletal health is well established and is the basis for the current Recommended Dietary Allowance. Vitamin D promotes calcium absorption in the small intestine and plays a significant role in calcium homeostasis and is important for mineralization of bone (Ross et al., 2011). Calcium also plays an important role in muscle, where the release of calcium triggers muscle contraction. Therefore, it is not surprising that vitamin D deficiency is associated with decreased muscle strength in older populations (Campbell & Allain, 2006). Vitamin D may also play an important role during muscle regeneration after injury. Srikuea and colleagues (2012) have shown that even though levels of the vitamin D receptor are low in normal adult muscle, during recovery from injury this receptor becomes highly expressed in the nuclei of regenerating fibers. This finding suggests that vitamin D plays an important role in muscle repair and it is not surprising that higher vitamin D levels tended to increase muscle hypertrophy following resistance exercise.
The results from older populations support the potential role of vitamin D in promoting musculoskeletal performance. However, in club-level athletes no significant improvements in bench press, leg press or vertical jump height were reported following 12 wk of vitamin D supplementation (20,000 or 40,000 IU/wk) (Close et al., 2013). The absence of a performance benefit occurred even though serum vitamin D (25(OH)D) levels increased significantly over the 12 wk study. Therefore, it is uncertain whether vitamin D use will improve performance in elite athletes.
Immune System
Vitamin D receptors are expressed on various immune cells (White, 2008). Vitamin D plays a role in both the innate immune response and the adaptive immune response and adequate levels of vitamin D are necessary for barrier integrity, production of antimicrobials, chemotaxis and regulation of the inflammatory response. The football season extends into the winter months, when vitamin D concentrations typically fall (Khare et al., 2013), and this may have a negative impact on the ability to fight a virus (Schwalfenberg, 2011). While our understanding of how vitamin D affects immune health is fairly new, the potential impact of keeping a player in the game vs. out with an illness is an important consideration when deciding on whether to supplement.
Sources of Vitamin D
Vitamin D is a fat-soluble vitamin, meaning that it requires dietary fat in order to be absorbed via the gut. There are a limited number of natural dietary sources of vitamin D, including fatty fish (tuna, salmon, mackerel, etc.), fish liver oil, cheese, egg yolks, and beef liver (Ross et al., 2011). Foods fortified with vitamin D, including milk, orange juice, yogurts, margarine and some cereals are also widely available (Ross et al., 2011).
Vitamin D can also be produced endogenously when the skin is exposed to ultraviolet B (UVB) rays. However, there are significant considerations as to whether sun-derived vitamin D will be sufficient for an athlete. In fall, winter and early spring at latitudes above 40o, the strength of the UVB rays is not enough for the skin to synthesize vitamin D. Further, with the colder temperatures, athletes typically wear additional layers of clothing thus blocking skin exposure. Athletes who spend significant time training and attending meetings indoors will have fewer opportunities to synthesize vitamin D regardless of time of year. The use of sunscreens also blocks the absorption of the UVB rays and inhibits the production of vitamin D. Skin color also plays a significant role as darker skin tones do not synthesize as much vitamin D as lighter skin tones. As a result of these limitations, most football players will not generate enough vitamin D on their own and need to consume sources via diet.
PRACTICAL NUTRITIONAL GUIDELINES BASED ON THE SCIENCE OF RECOVERY
-
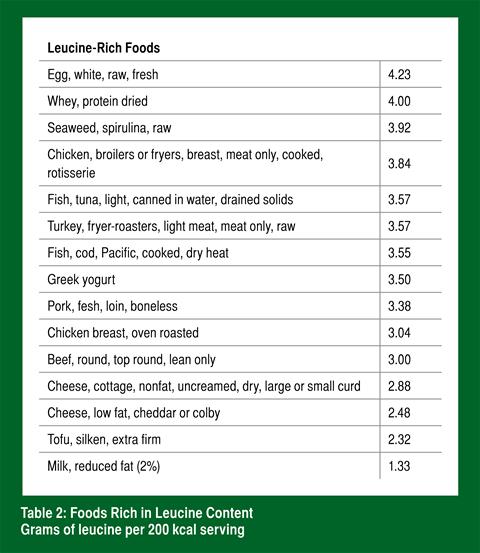
Immediately following games and training, consume a rapidly absorbable protein source rich in the amino acid leucine. Examples of these types of food are found in Table 2 and include dairy products (specifically the whey component) and eggs.
-
Throughout the season, eat meals containing 0.25 g/kg body mass of leucine-rich protein first thing in the morning and then every 4 h throughout the day (Moore et al., 2009). Eating fewer meals with more protein is not as good as eating this optimal amount (Areta et al., 2013).
-
Consume 0.5 g/kg body mass of leucine-rich protein right before bed. This improves protein synthesis while sleeping and maintains a positive protein balance overnight (Res et al., 2012).
-
Athletes should consume ~3 g of Omega-3 fatty acids/d with ~0.75 g of EPA and 1g DHA. Ideally, this amount of Omega-3 fatty acids would be consumed in whole foods such as tuna and salmon, flax seeds, nuts and nut butters. This amount of Omega-3 fats may assist the athlete by increasing muscle protein synthesis, decreasing inflammation and improving cognitive function.
-
Athletes who have documented vitamin D insufficiency by blood test should consume ~5,000 IU of vitamin D/day. Further, for African American players or those who practice and live further north and may not produce as much vitamin D in the skin, a similar supplementation program may decrease infections and improve muscle recovery.
REFERENCES
Areta, J. L., L. M. Burke, M. L. Ross, D. M. Camera, D. W. West, E. M. Broad, N. A. Jeacocke, D. R. Moore, T. Stellingwerff, S. M. Phillips, J. A. Hawley, and V. G. Coffey (2013). Timing and distribution of protein ingestion during prolonged recovery from resistance exercise alters myofibrillar protein synthesis. J Physiol. 591: 2319-2331.
Baar, K., and K. Esser (1999). Phosphorylation of p70(S6k) correlates with increased skeletal muscle mass following resistance exercise. Am. J. Physiol. 276: C120-C127.
Breen, L., and T. A. Churchward-Venne (2012). Leucine: a nutrient 'trigger' for muscle anabolism, but what more? J. Physiol. 590: 2065-2066.
Burdge, G. C., A. E. Jones, and S. A. Wootton (2002). Eicosapentaenoic and docosapentaenoic acids are the principal products of alpha-linolenic acid metabolism in young men. Br. J. Nutr. 88: 355-363.
Calder, P. C. (2006). N-3 polyunsaturated fatty acids, inflammation, and inflammatory diseases. Am. J. Clin. Nutr. 83: 1505S-1519S.
Campbell, P. M., and T. J. Allain (2006). Muscle strength and vitamin D in older people. Gerontology 52: 335-338.
Churchward-Venne, T. A., N. A. Burd, C. J. Mitchell, D. W. West, A. Philp, G. R. Marcotte, S. K. Baker, K. Baar, and S. M. Phillips (2012). Supplementation of a suboptimal protein dose with leucine or essential amino acids: effects on myofibrillar protein synthesis at rest and following resistance exercise in men. J.Physiol 590: 2751-2765.
Close, G. L., J. Leckey, M. Patterson, W. Bradley, D. J. Owens, W. D. Fraser, and J. P. Morton (2013). The effects of vitamin D(3) supplementation on serum total 25[OH] D concentration and physical performance: a randomised dose-response study. Br J. Sports Med. 47: 692-696.
Coffey, V. G., B. Jemiolo, J. Edge, A. P. Garnham, S. W. Trappe, and J. A. Hawley (2009). Effect of consecutive repeated sprint and resistance exercise bouts on acute adaptive responses in human skeletal muscle. Am. J. Physiol. 297: R1441-R1451.
Dickinson, J. M., C. S. Fry, M. J. Drummond, D. M. Gundermann, D. K. Walker, E. L. Glynn, K. L. Timmerman, S. Dhanani, E. Volpi, and B. B. Rasmussen (2011). Mammalian target of rapamycin complex 1 activation is required for the stimulation of human skeletal muscle protein synthesis by essential amino acids. J. Nutr. 141: 856-862.
Drummond, M. J., C. S. Fry, E. L. Glynn, H. C. Dreyer, S. Dhanani, K. L. Timmerman, E. Volpi, and B. B. Rasmussen (2009). Rapamycin administration in humans blocks the contraction-induced increase in skeletal muscle protein synthesis. J. Physiol. 587: 1535-1546.
Dyall, S. C., and A. T. Michael-Titus (2008). Neurological benefits of Omega-3 fatty acids. Neuromol. Med .10: 219-235.
Farrokhyar, F., R. Tabasinejad, D. Dao, D. Peterson, O. R. Ayeni, R. Hadioonzadeh, and M. Bhandari (2015). Prevalence of Vitamin D Inadequacy in Athletes: A Systematic- Review and Meta-Analysis. Sports Med. 45:365-378.
Fontani, G., F. Corradeschi, A. Felici, F. Alfatti, S. Migliorini, and L. Lodi (2005). Cognitive and physiological effects of Omega-3 polyunsaturated fatty acid supplementation in healthy subjects. Eur J Clin Invest 35: 691-699.
Ge, Y., A. L. Wu, C. Warnes, J. Liu, C. Zhang, H. Kawasome, N. Terada, M. D. Boppart, C. J. Schoenherr, and J. Chen (2009). mTOR regulates skeletal muscle regeneration in vivo through kinase-dependent and kinase-independent mechanisms. Am. J. Physiol. 297: C1434-C1444.
Gogus, U. S. (2010). n-3 Omega fatty acids: a review of current knowledge. Int. J. Food Sci. Tech. 45: 417-436.
Gray, B. L., J. M. Buchowski, D. B. Bumpass, R. A. Lehman, Jr., N. A. Mall, and M. J. Matava (2013). Disc herniations in the National Football League. Spine 38: 1934- 1938.
Hamilton, D. L., A. Philp, M. G. MacKenzie, A. Patton, M. C. Towler, I. J. Gallagher, S. C. Bodine, and K. Baar (2014). Molecular brakes regulating mTORC1 activation in skeletal muscle following synergist ablation. Am. J. Physiol. 307: E365-E373.
Hwee, D. T., L. M. Baehr, A. Philp, K. Baar, and S. C. Bodine (2014). Maintenance of muscle mass and load-induced growth in Muscle RING Finger 1 null mice with age. Aging Cell 13: 92-101.
Jouris, K. B., J. L. McDaniel, and E. P. Weiss (2011). The effect of Omega-3 fatty acid supplementation on the inflammatory response to eccentric strength exercise. J. Sports Sci. Med. 10: 432-438.
Khare, D., N. M. Godbole, S. D. Pawar, V. Mohan, G. Pandey, S. Gupta, D. Kumar, T. N. Dhole, and M. M. Godbole (2013). Calcitriol [1, 25[OH]2 D3] pre- and post-treatment suppresses inflammatory response to influenza A (H1N1) infection in human lung A549 epithelial cells. Eur. J. Nutr. 52:1405-1415.
Koh, T. J. and F. X. Pizza (2009). Do inflammatory cells influence skeletal muscle hypertrophy? Front. Biosci. 1: 60-71.
Kraemer, W. J., D. P. Looney, G. J. Martin, N. A. Ratamess, J. L. Vingren, D. N. French, D. L. Hatfield, M. S. Fragala, B. A. Spiering, R. L. Howard, C. Cortis, T. K. Szivak, B. A. Comstock, C. Dunn-Lewis, D. R. Hooper, S. D. Flanagan, J. S. Volek, J. M. Anderson, C. M. Maresh, and S. J. Fleck (2013). Changes in creatine kinase and cortisol in National Collegiate Athletic Association Division I American football players during a season. J. Strength Cond. Res. 27: 434-441.
Kris-Etherton, P. M., W. S. Harris, and L. J. Appel (2002). Fish consumption, fish oil, Omega-3 fatty acids, and cardiovascular disease. Circulation 106: 2747-2757.
Mall, N. A., J. Buchowski, L. Zebala, R. H. Brophy, R. W. Wright and, M. J. Matava (2012). Spine and axial skeleton injuries in the National Football League. Am. J. Sports Med. 40: 1755-1761.
Maroon, J. C., C. M. Mathyssek, J. W. Bost, A. Amos, R. Winkelman, A. P. Yates, M. A. Duca, and J. A. Norwig (2015). "Vitamin D profile in National Football League players. Am J Sports Med. E-pub ahead of print. PMID # 25649084.
Moberg, M., W. Apro, I. Ohlsson, M. Ponten, A. Villanueva, B. Ekblom, and E. Blomstrand (2014). Absence of leucine in an essential amino acid supplement reduces activation of mTORC1 signalling following resistance exercise in young females. Appl. Physiol. Nutr. Metab. 39: 183-194.
Moore, D. R., M. J. Robinson, J. L. Fry, J. E. Tang, E. I. Glover, S. B. Wilkinson, T. Prior, M. A. Tarnopolsky, and S. M. Phillips (2009). Ingested protein dose response of muscle and albumin protein synthesis after resistance exercise in young men. Am. J. Clin. Nutr. 89: 161-168.
Parr, E. B., D. M. Camera, J. L. Areta, L. M. Burke, S. M. Phillips, J. A. Hawley, and V. G. Coffey (2014). Alcohol ingestion impairs maximal post-exercise rates of myofibrillar protein synthesis following a single bout of concurrent training. PLoS One 9: e88384.
Pasiakos, S. M., L. M. Margolis, and J. S. Orr (2014). Optimized dietary strategies to protect skeletal muscle mass during periods of unavoidable energy deficit. Faseb J. E-pub ahead of print. PMID # 25550460.
Phillips, S. M., K. D. Tipton, A. Aarsland, S. E. Wolf, and R. R. Wolfe (1997). Mixed muscle protein synthesis and breakdown after resistance exercise in humans. Am. J. Physiol. 273: E99-E107.
Phillips, S. M., K. D. Tipton, A. A. Ferrando, and R. R. Wolfe (1999). Resistance training reduces the acute exercise-induced increase in muscle protein turnover. Am. J. Physiol. 276: E118-E124.
Res, P. T., B. Groen, B. Pennings, M. Beelen, G. A. Wallis, A. P. Gijsen, J. M. Senden, and L. van Loon (2012). Protein ingestion before sleep improves postexercise overnight recovery. Med. Sci. Sports Exerc 44: 1560-1569.
Ross, A. C., J. E. Manson, S. A. Abrams, J. F. Aloia, P. M. Brannon, S. K. Clinton, R. A. Durazo-Arvizu, J. C. Gallagher, R. L. Gallo, G. Jones, C. S. Kovacs, S. T. Mayne, C. J. Rosen, and S. A. Shapses (2011). The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: what clinicians need to know. J. Clin. Endocrinol. Metab. 96: 53-58.
Schwalfenberg, G. K. (2011). A review of the critical role of vitamin D in the functioning of the immune system and the clinical implications of vitamin D deficiency. Mol. Nutr. Food Res. 55: 96-108.
Smith, G. I., P. Atherton, D. N. Reeds, B. S. Mohammed, D. Rankin, M. J. Rennie, and B. Mittendorfer (2011a). Dietary Omega-3 fatty acid supplementation increases the rate of muscle protein synthesis in older adults: a randomized controlled trial. Am. J. Clin. Nutr. 93: 402-412.
Smith, G. I., P. Atherton, D. N. Reeds, B. S. Mohammed, D. Rankin, M. J. Rennie, and B. Mittendorfer (2011b). Omega-3 polyunsaturated fatty acids augment the muscle protein anabolic response to hyperinsulinaemia-hyperaminoacidaemia in healthy young and middle-aged men and women. Clin. Sci. 121: 267-278.
Srikuea, R., X. Zhang, O. K. Park-Sarge, and K. A. Esser (2012). VDR and CYP27B1 are expressed in C2C12 cells and regenerating skeletal muscle: potential role in suppression of myoblast proliferation. Am. J. Physiol. 303: C396-C405.
Tartibian, B., B. H. Maleki, and A. Abbasi (2009). The effects of ingestion of Omega-3 fatty acids on perceived pain and external symptoms of delayed onset muscle soreness in untrained men. Clin. J. Sport Med. 19: 115-119.
Tipton, K. D., A. A. Ferrando, S. M. Phillips, D. Doyle, Jr., and R. R. Wolfe (1999). Postexercise net protein synthesis in human muscle from orally administered amino acids. Am. J. Physiol. 276:E628-E634.
White, J. H. (2008). Vitamin D signaling, infectious diseases, and regulation of innate immunity. Infect. Immun. 76: 3837-3843.
Zimmerman, J. L., and M. C. Shen (2013). Rhabdomyolysis. Chest 144: 1058-1065.