High-Intensity Interval Training and the Impact of Diet

KEY POINTS
-
While most studies have focused on traditional aerobic/endurance or strength/resistance exercise, dietary interventions can also alter the acute and chronic responses to interval-type exercise.
-
It is difficult to draw firm conclusions regarding the effect of specific dietary manipulations on interval training adaptations as findings from limited studies, which vary with respect to subject training status and the nature of the exercise and nutritional interventions, have been equivocal.
-
Restricting carbohydrate availability alters the acute exercise-induced activation of specific nutrient-sensitive signaling molecules, and periodic training using this practice may augment gains in mitochondrial content and possibly, exercise capacity.
-
Sodium bicarbonate ingestion improves acute high-intensity exercise capacity and may augment training-induced improvements in performance, with limited evidence suggesting this adaptation may be related to an enhanced capacity for mitochondrial respiration.
-
Beta-alanine supplementation increases skeletal muscle carnosine content and enhances performance during acute high-intensity exercise, but does not augment skeletal muscle or performance adaptations to interval training.
INTRODUCTION
High-intensity interval training generally refers to repeated bouts of relatively intense exercise interspersed by periods of rest or lower intensity exercise for recovery. It has long been appreciated by athletes that “intervals” are an effective means to enhance aerobic energy provision when added into a well-structured training program, although the optimal “mix” of intensity vs. volume for performance remains debatable (Laursen, 2010). A resurgence of interest in the topic of interval training over the last decade has advanced our understanding of the physiological mechanisms by which interval training enhances aerobic energy provision in normal healthy individuals as well as those at risk or afflicted by chronic diseases (Gibala et al., 2012; Gibala & Jones, 2013; Weston et al., 2014). One of the most striking findings of this research has been that interval training per se is a potent stimulus to induce physiological adaptations that resemble changes typically associated with traditional endurance training, despite a lower total exercise volume and reduced training time commitment (Gibala et al., 2012; Gibala et al., 2014).
With respect to nomenclature, and given the wide array of interval training protocols and descriptors, a classification scheme was recently proposed in order to try and standardize terminology in the literature (Weston et al., 2014). It was suggested that “high-intensity interval training” (HIIT) be used to describe protocols in which the training stimulus is “near maximal” or the target intensity is between 80-100% of maximal heart rate, whereas “sprint interval training” be used to describe protocols that involve “all out” or “supramaximal” efforts, in which target intensities correspond to workloads greater than what is required to elicit peak or maximal oxygen uptake (Weston et al., 2014). However, for simplicity and given the widespread use of the acronym in athletic circles, the term HIIT will be used exclusively herein, along with clear descriptions of the training interventions employed in various studies.
The purpose of this brief review is to consider the potential for dietary interventions, either in the form of altered macronutrient intake or the ingestion of specific nutritional supplements, to alter the adaptive response to interval training. While it is well established that specific nutritional strategies can modulate the acute and chronic responses to training, most studies have focused on traditional aerobic/endurance or strength/resistance exercise (Hawley et al., 2011). Dietary interventions can also alter the response to interval-type exercise, and the emphasis here will be placed on recent studies that have examined training responses under conditions of reduced carbohydrate availability and the impact of bicarbonate ingestion and beta-alanine supplementation on training responses.
REDUCED CARBOHYDRATE AVAILABILITY
Athletes are typically advised to habitually consume a diet high in carbohydrates, since this is the major macronutrient utilized for energy during high-intensity exercise. Paradoxically, as recently reviewed by Bartlett et al. (2015), commencing at least some training sessions with reduced carbohydrate availability has been reported to augment the physiological adaptations to training. The theoretical basis for this concept is that glycogen is more than a substrate for energy, but rather its availability in skeletal muscle (i.e., a relative lack or surplus) can regulate the adaptive response to exercise (Philp et al., 2012). It is also possible that manipulating carbohydrate availability alters other metabolic intermediates and thus could induce adaptations that are independent of changes in glycogen content per se (Baar, 2014).
With respect to studies that have specifically examined whether manipulating carbohydrate availability influences the adaptive response to interval training, researchers have acutely altered macronutrient intake prior to exercise (Bartlett et al., 2013) and also during recovery between twice-daily training sessions (Cochran et al., 2010). Commencing exercise with reduced muscle glycogen content (Bartlett et al., 2013), or restricting carbohydrate intake prior to a second daily bout of exercise (Cochran et al., 2010), has been shown to augment the acute activation of molecular signaling cascades linked to skeletal muscle remodeling during recovery. For example, Cochran et al. (2010) studied 10 men who performed two trials in random order separated by 1 wk. Each trial consisted of a morning and afternoon training session, both of which involved 5 x 4 min cycling at ~90-95% of heart rate reserve. The trials were separated by 3 h of recovery during which subjects ingested a high-carbohydrate drink (HI-HI) or a non-energetic placebo (HI-LO) before the afternoon training session. Biopsies revealed that muscle phosphocreatine and adenosine triphosphate (ATP) content were similar after the morning exercise but decreased to a greater extent after the afternoon training session in HI-LO vs. HI-HI. This was associated with greater phosphorylation of p38 mitogen-activated protein kinase, although there were no differences between treatments in the expression of another important protein that regulates mitochondrial biogenesis, peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α). Using a different experimental design, Bartlett et al. (2013) also demonstrated that commencing interval exercise with reduced muscle glycogen content increased the phosphorylation of the regulatory protein p53, although similar to Cochran et al. (2010), this was not associated with changes in the exercise-induced expression of PGC-1α. While a host of metabolic intermediates may be at play (Baar, 2014), these acute studies provide evidence of nutrient-sensitive signaling molecules that could potentially alter the chronic response to training under conditions of restricted carbohydrate intake.
Several studies have reported that chronically manipulating carbohydrate availability over the course of an interval-training program alters the adaptive response of skeletal muscle (for review, see Bartlett et al., 2015). For example, Morton et al. (2009) had three groups of recreationally active men perform high-intensity intermittent running four times/wk for 6 wk. Groups 1 and 2 trained twice daily, 2 days/wk, and ingested either a glucose drink or a placebo, at regular intervals during the second daily session. The third group trained once/day, 4 days/wk and consumed no beverage during exercise. Training increased mitochondrial content in all groups, as evidenced by increased succinate dehydrogenase maximal activity, but the increase was greatest in the subjects who trained twice daily and received no carbohydrate (Group 2). The authors concluded that training under conditions of reduced carbohydrate availability provided an enhanced stimulus for inducing oxidative enzyme adaptations of skeletal muscle, although this did not translate into improved performance during high-intensity exercise. These results are generally consistent with the majority of other studies that have manipulated carbohydrate availability through overnight fasting and/or manipulating carbohydrate feeding between twice daily exercise sessions, and reported no differences in exercise performance across groups (Bartlett et al., 2015).
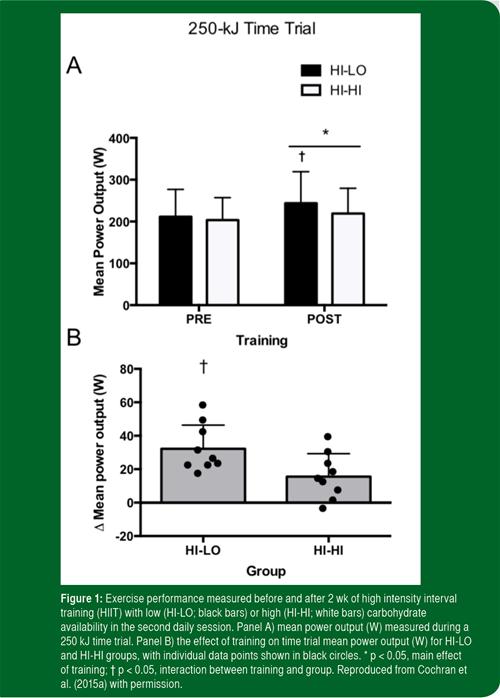
It has been suggested that short blocks of “training low” might be more practical and beneficial than longer interventions, since prolonged periods of training under conditions of reduced carbohydrate availability are potentially detrimental because of compromised immune function and increased muscle protein breakdown. Cochran et al. (2015a) recently examined whether restricting carbohydrate intake between twice daily sessions of interval exercise over a period of 2 wk would augment improvements in exercise performance and mitochondrial content. Using a design similar to the acute study performed by the same group of investigators (Cochran et al., 2010), 18 active but not highly trained subjects were matched for age, sex and fitness and randomly allocated to one of two groups. On each of six training days over 2 wk, subjects completed two training sessions separated by 3 h. Each session involved 5 x 4 min cycling intervals at an intensity of 60% of peak power output elicited during the VO2peak test, interspersed by 2 min of recovery. Subjects ingested either ~200 g of carbohydrate (HI-HI) or <20 g of CHO (HI-LO) during the 3 h period between bouts. The most intriguing finding was that the training-induced improvement in 250 kJ cycling time trial performance was greater in the HI-LO group compared to the HI-HI group (Figure 1). The mechanism for the superior performance adaptation was not readily apparent, as the training-induced increases in enzymatic markers of mitochondrial content were not different between groups. While further research is needed to determine whether more highly-trained individuals respond in a similar manner, the study by Cochran et al. (2015a) is the first to show that a short-term “train low, compete high” training intervention can improve whole-body exercise capacity.
BICARBONATE INGESTION
Bicarbonate is an extracellular ion that plays a role in maintaining blood pH. The ingestion of large amounts of bicarbonate transiently increases blood bicarbonate and pH, resulting in an enhanced capacity to buffer changes in cellular acidity under conditions of increased hydrogen ion accumulation, e.g., during strenuous exercise. While the precise role of pH in muscle fatigue is unclear, it is well established that increasing buffering capacity through bicarbonate loading can acutely enhance high-intensity exercise performance (Burke 2013). Ingestion of sodium bicarbonate (NaHCO3) is a common practice in this regard, and a meta-analysis concluded that ingestion of a dose equivalent to ~0.3 g/kg body mass improved performance during a single 60 s sprint by ~2%, with a further ~1% modifying effect when sprints are repeated (Carr et al., 2011). Combining chronic sodium bicarbonate ingestion with interval training is potentially beneficial because the capacity to produce work is elevated, which may in turn lead to a greater cumulative training stimulus.
There is also evidence that sodium bicarbonate supplementation per se favorably alters the metabolic response to HIIT such that adaptations are enhanced even if the training stimulus is held constant. Edge et al. (2006) studied 16 moderately trained women who performed 24 sessions of interval training over 8 wk. Each session involved 6-12, 2 min cycle intervals at 140-170% of their lactate threshold, and total work was matched between groups. In a blinded manner, subjects ingested either sodium bicarbonate or a placebo before each training session. The training-induced increase in lactate threshold and time to fatigue during exercise at an intensity equivalent to 100% of pre-training VO2peak was higher in the sodium bicarbonate group compared to placebo. The authors speculated that bicarbonate ingestion may have reduced metabolic acidosis during training or stimulated a greater improvement in muscle oxidative capacity (Edge et al., 2006).
In support of this theory, Bishop et al. (2010) reported that mitochondrial respiratory capacity in rats was increased to a greater extent when HIIT was supplemented with sodium bicarbonate as compared to a placebo, and was associated with a 52% greater increase in running time to exhaustion in the sodium bicarbonate group. Male rats were randomly assigned to either a control, placebo or sodium bicarbonate group. The former served as a sedentary control, whereas the other two groups ingested either water or sodium bicarbonate 30 min prior to training, which consisted of 7-12, 2 min running intervals performed 5 times/wk for 5 wk. Compared to control, as well as the placebo treatment, the training-induced improvement in markers of mitochondrial mass and mitochondrial respiration were increased to a greater extent in the sodium bicarbonate group.
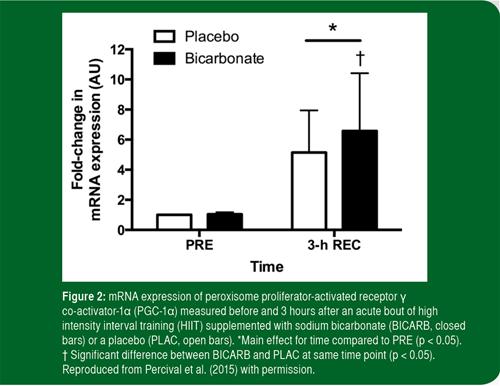
While no training studies in humans have attempted to replicate the findings of Bishop et al. (2010), Percival et al. (2015) recently tested the hypothesis that ingestion of sodium bicarbonate prior to an acute session of interval training would augment signaling cascades and gene expression linked to mitochondrial biogenesis in human skeletal muscle. On two occasions separated by 1 wk, active but not highly-trained men performed a HIIT protocol that involved 10, 60 s cycling efforts at an intensity eliciting ~90% of maximal heart rate, interspersed with 60 s of recovery. In a double-blind, crossover manner, subjects ingested a total of 0.4 g/kg body mass of sodium bicarbonate, administered in two half-doses at 90 and 60 min prior to exercise, or an equimolar amount of the placebo, sodium chloride. Venous blood samples confirmed that bicarbonate and pH were elevated at all time points post-ingestion in the bicarbonate supplemented condition, and this was associated with greater muscle glycogen utilization and blood lactate accumulation during exercise. The acute exercise-induced increase in the phosphorylation of acetyl-CoA carboxylase, a downstream marker of AMP-activated protein kinase activity, and p38 mitogen-activated protein kinase were similar between treatments. However, the increase in PGC-1α mRNA expression after 3 h of recovery was higher (7 fold above rest) after sodium bicarbonate ingestion compared to placebo (5 fold above rest) (Figure 2). The authors concluded that bicarbonate ingestion altered the expression of this key regulatory protein associated with mitochondrial biogenesis. The elevated PGC-1α mRNA response provides a putative mechanism to explain the enhanced mitochondrial adaptation seen after chronic interval training combined with bicarbonate supplementation in rats.
BETA-ALANINE SUPPLEMENTATION
Beta-alanine (ß-alanine) is a non-proteinogenic amino acid and the rate-determining precursor for the synthesis of carnosine, a dipeptide formed with histidine that functions as a physiologically relevant pH buffer in skeletal muscle (Harris & Stellingwerff, 2013). It is now well established that ß-alanine supplementation can increase carnosine content in human skeletal muscle, with numerous factors influencing this process, including the dose and duration of supplementation as well as meal composition and training status (Blancquaert et al., 2015). Many studies have examined the potential for ß-alanine to enhance acute exercise performance, and a meta-analysis (Hobson et al., 2012) concluded that ß-alanine is beneficial for exercise lasting ≥1 min, i.e., a bout duration that is characteristic of many types of interval training protocols. While the same meta-analysis determined the effect was absent for shorter exercise durations, it has been subsequently demonstrated that ß-alanine enhances performance during intermittent, or repeated-type, exercise in which the high-intensity bout duration is <60 sec (Blancquaert et al., 2015).
Given the potential for ß-alanine supplementation to enhance acute high-intensity exercise performance (Blancquaert et al., 2015), investigators have considered the potential for this practice to augment adaptations to interval training (Cochran et al., 2015b; Gross et al., 2014). Gross et al. (2014) had 16 active men ingest either 3.2 g/d of ß-alanine or a dextrose placebo for 38 d (n=8 per group), with supplementation then discontinued prior to an 11 d block of interval training. The program involved a total of 9 sessions, each consisting of a 10 min warm-up, followed by 4 x 4 min bouts of cycling at an intensity that elicited ~90-95% of maximal heart rate, interspersed by 3 min of light cycling for recovery. A battery of metabolic and exercise tolerance tests were performed at three time points: before and after supplementation and following the training intervention. Magnetic resonance spectroscopy confirmed that the ß-alanine supplementation protocol significantly increased the leg muscle content of carnosine by ~32% prior to training, although there was considerable variability in this response. ß-alanine supplementation was associated with an increased aerobic energy contribution during a test that involved 90 s of intense cycling at ~110% of peak power output (relative to VO2peak), compared to pre-supplementation. Despite the increase in muscle carnosine content after ß-alanine supplementation, there were no between-group differences in training adaptations. Training increased buffering capacity measured in biopsy samples from the vastus lateralis muscle, but there was no change in mitochondrial markers of oxidative capacity or aerobic energy metabolism during intense cycling exercise.
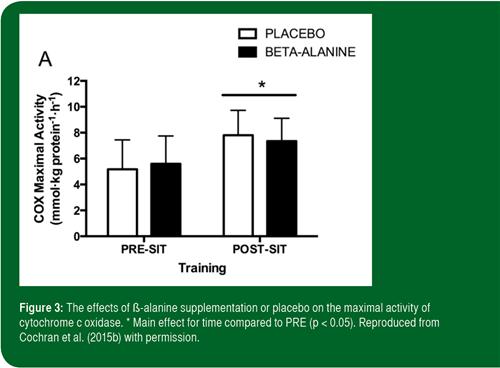
Cochran et al. (2015b) also examined the effect of ß-alanine supplementation on skeletal muscle and performance adaptations to interval training in young active men. Using a randomized, double-blind design similar to Gross et al. (2014), the study involved a daily dose of 3.2 g of ß-alanine or a dextrose placebo. However, a key difference was that following a 4 wk period of baseline supplementation, subjects continued to ingest either ß-alanine or placebo capsules over the course of a 6 wk training intervention. Each of three weekly sessions consisted of 4-6 Wingate Tests, i.e., 30 s bouts of “all out” maximal cycling, interspersed with 4 min of light cycling for recovery. Before and after the training program, subjects completed a 250 kJ time trial and a repeated sprint test. Biopsies from the vastus lateralis muscle revealed that muscle carnosine content increased by ~33 and ~52% after 4 and 10 wk of β-alanine supplementation, but was unchanged in the placebo condition. Total work done during training increased over the interval training protocol, but the increase was similar across treatments in spite of the differences in muscle carnosine content. Similarly, while interval training increased markers of mitochondrial content (Figure 3), as well as VO2peak, repeated-sprint capacity, and 250 kJ time trial performance, there were no differences between treatments. The authors theorized that the sprint-training stimulus may have overwhelmed any potential influence of β-alanine, or the supplementation protocol was insufficient to measurably alter the variables investigated. In summary, based on a limited number of studies conducted to date, there is little evidence at present to suggest that β-alanine supplementation enhances physiological or performance adaptations to interval training.
SUMMARY AND PRACTICAL APPLICATIONS
Numerous dietary interventions could in theory augment the adaptive response to interval training, either by 1) improving energy metabolism during exercise, which could facilitate greater total work and an enhanced chronic training stimulus, or 2) promoting some aspect of the adaptive response during recovery, which could lead to enhanced physiological adaptations over time. In practice, equivocal findings from limited studies, which vary with respect to subject training status and the nature of the exercise and nutritional interventions employed, make it difficult to draw firm conclusions regarding the effect of specific dietary manipulations on interval training adaptations. This limitation is reinforced by the inherent difficulty in extrapolating laboratory-derived measurements to the typical competitive environment of the athlete.
With respect to the specific interventions reviewed here, there are data to suggest that restricting carbohydrate availability acutely alters the exercise-induced activation of specific nutrient-sensitive signaling molecules, and some evidence suggests that periodic training in this manner can augment gains in mitochondrial content and possibly, exercise capacity. Sodium bicarbonate ingestion improves acute high-intensity exercise capacity and may augment training-induced improvements in performance, with limited evidence suggesting that this adaptation may be related to an enhanced capacity for mitochondrial respiration. Finally, beta-alanine supplementation increases skeletal muscle carnosine content and enhances performance during acute high-intensity exercise, but does not appear to augment skeletal muscle or performance adaptations to interval training.
REFERENCES
Baar, K. (2014). Nutrition and the adaptation to endurance training. Sports Med. 44:S5-12.
Bartlett, J.D., J. Louhelainen, Z. Iqbal, A.J. Cochran, M.J. Gibala, W. Gregson, G.L. Close, B. Drust, and J.P. Morton (2013). Reduced carbohydrate availability enhances exercise-induced p53 signaling in human skeletal muscle: implications for mitochondrial biogenesis. Am. J. Physiol. 304:R450-458.
Bartlett, J.D., J.A. Hawley, and J.P. Morton (2015). Carbohydrate availability and exercise training adaptation: Too much of a good thing? Eur. J. Sport Sci. 15:3-12.
Bishop, D.J., C. Thomas, T. Moore-Morris, M. Tonkonogi, K. Sahlin, and J. Mercier (2010). Sodium bicarbonate ingestion prior to training improves mitochondrial adaptations in rats. Am. J. Physiol. 299: E225-233.
Blancquaert, L., I. Everaert, and W. Derave (2015). Beta-alanine supplementation, muscle carnosine and exercise performance. Curr. Opin. Clin. Nutr. Metab. Care. 18:63-70.
Burke, L.M. (2013). Practical considerations for bicarbonate loading and sports performance. Nestle Nutr. Inst. Workshop Ser. 75:15-26.
Carr, A.J., W.G. Hopkins, and C.J. Gore (2011). Effects of acute alkalosis and acidosis on performance: a meta-analysis. Sports Med. 2011 41:801-814.
Cochran, A.J., J.P. Little, M.A. Tarnopolsky, and M.J. Gibala (2010). Carbohydrate feeding during recovery alters the skeletal muscle metabolic response to repeated sessions of high-intensity interval exercise in humans. J. Appl. Physiol. 108:628-636.
Cochran, A.J.C., F. Myslik, M.J. MacInnis, M.E. Percival, D. Bishop, M. Tarnopolsky, and M.J. Gibala (2015a). Manipulating carbohydrate availability between twice-daily sessions of high-intensity interval training over two weeks improves time-trial performance. Int. J. Sports Nutr. Exerc. Metab. Mar 26, E-pub, PMID 25811132.
Cochran, A.J.R., M.E. Percival, S. Thompson, J.B. Gillen, M.J. MacInnis, M.A. Tarnopolsky, M. Potter, and M.J. Gibala (2015b). Beta-alanine supplementation does not augment the skeletal muscle adaptive response to six weeks of sprint-interval training. Int. J. Sports Nutr. Exerc. Metab. May 22, E-pub, PMID 26008634.
Edge, J., Bishop, D., and C. Goodman (2006). Effects of chronic NaHCO3 ingestion during interval training on changes to muscle buffer capacity, metabolism, and short-term endurance performance. J. Appl. Physiol. 101:918-925.
Gibala, M.J., J.P. Little, M.J. MacDonald, and J.A. Hawley (2012). Physiological adaptations to low-volume, high-intensity interval training in health and disease. J. Physiol. 590:1077-1084.
Gibala, M.J., and A.M. Jones (2013). Physiological and performance adaptations to high-intensity interval training. Nestle Nutr. Inst. Workshop Ser. 76:51-60.
Gibala, M.J., J.B. Gillen, and M.E. Percival (2014). Physiological and health-related adaptations to low-volume interval training: influences of nutrition and sex. Sports Med. 44:S127-137.
Gross, M., C. Boesch, C.S. Bolliger, B. Norman, T. Gustafsson, H. Hoppeler, and M. Vogt. (2014). Effects of beta-alanine supplementation and interval training on physiological determinants of severe exercise performance. Eur. J. Appl. Physiol. 114:221-234.
Harris, R.C., and T. Stellingwerff (2013). Effect of β-alanine supplementation on high-intensity exercise performance. Nestle Nutr. Inst. Workshop Ser. 76:61-71.
Hawley, J.A., L.M. Burke, S.M. Phillips, and L.L. Spriet (2011). Nutritional modulation of training-induced skeletal muscle adaptations. J. Appl. Physiol. 110:834-845.
Hobson, R.M., B. Saunders, G. Ball, R.C. Harris, and C. Sale (2012). Effects of β-alanine supplementation on exercise performance: a meta-analysis. Amino Acids. 43:25-37.
Laursen, P.B. (2010). Training for intense exercise performance: high-intensity or high-volume training? Scand. J. Med. Sci. Sports. 20:S1-10.
Morton, J.P., L. Croft, J.D. Bartlett, D.P. Maclaren, T. Reilly, L. Evans, A. McArdle, and B. Drust (2009). Reduced carbohydrate availability does not modulate training-induced heat shock protein adaptations but does upregulate oxidative enzyme activity in human skeletal muscle. J. Appl. Physiol. 106:1513-1521.
Percival, M.E., M.J. Martin, J.B. Gillen, L.E. Skelly, M.J. MacInnis, A.E. Green, M.A. Tarnopolsky, and M.J. Gibala (2015). Sodium bicarbonate ingestion augments the increase in PGC-1α mRNA expression during recovery from intense interval exercise in human skeletal muscle. J Appl Physiol Sept 17, E-pub, PMID 26384407.
Philp, A., M. Hargreaves, and K. Baar (2012). More than a store: regulatory roles for glycogen in skeletal muscle adaptation to exercise. Am. J. Physiol. 302:E1343-1351.
Weston, K.S., U. Wisløff, and J.S. Coombes (2014). High-intensity interval training in patients with lifestyle-induced cardiometabolic disease: a systematic review and meta-analysis. Br. J. Sports Med. 48:1227-1234.








