Exercise, Nutrition and the Brain

KEY POINTS
- Exercise and nutrition can influence brain development in youth.
- Exercise and nutrition can slow down and reverse cognitive decline in the elderly.
- Exercise and nutrition influences brain health through several mechanisms that create new neurons (neurogenesis).
- Fatigue during exercise can reside in the brain.
- Nutritional interventions may influence the occurrence of “central or brain fatigue” during exercise.
INTRODUCTION
Physical activity has been associated with the reduction of a number of physical and mental disorders. There is now ample evidence that physical activity will decrease the incidence of cardiovascular disease, colon and breast cancer and obesity, but also diseases such as Alzheimer’s, depression and anxiety (Gómez-Pinilla, 2011; Van Praag, 2009). A number of large, prospective and cross-sectional observational studies find that the dietary profile benefiting cognitive function with aging contains weekly servings (2 - 5) of fish and multiple daily servings of cereals, darkly or brightly colored fruits and leafy vegetables (Parrott & Greenwood, 2007). Both diet and exercise have therefore been used as interventions to reverse the possible negative effect of ageing in brain function. This paper will describe how exercise and nutrition can influence brain development, brain performance and cognition (e.g., remembering things, like where did I park my car at the airport?).
THE VALUE OF EXERCISE AND NUTRITION FOR THE BRAIN
Children - The Developing Brain
There are strong indications that children are growing increasingly sedentary and unfit, and that these lifestyle factors are related to an earlier onset of several chronic diseases such as type 2 diabetes and obesity. Several cross-sectional and longitudinal studies have provided proof for an association between being overweight and poor academic performance. Aerobic fitness has also been positively linked with cognition and academic achievement (Davis & Cooper, 2011). Several studies suggest that children’s cognitive ability and school performance may be affected by their general physical condition (Davis & Cooper, 2011).
Diet can also substantially influence both the development and health of brain structure and function. A healthy diet provides building blocks for the brain to create and maintain neoronal connections, which are critical for improved cognition and academic performance. Dietary factors have a broad and positive action on neuronal function and plasticity. For example, the omega-3 fatty acids provide building material for the brain. They are essential for supporting intercellular signaling events, and therefore, positively influence synaptic function. However, diets rich in sugar or saturated fats or high in calories are considered deleterious for neural function, as they act to elevate levels of oxidative stress and to reduce synaptic plasticity and cognitive functions (Gómez-Pinilla, 2011). Brain function is certainly dependent on adequate nutrition, and short-term variations in the amount and composition of nutrient intake in healthy individuals influence measures of cognitive function. Studies have shown that eating breakfast is associated with several positive effects on the cognitive functioning of well-nourished children (Hoyland et al., 2009; Taki et al., 2010). Exercise has been shown to interact with dietary interventions — increasing the positive effects on brain functioning and decreasing the unhealthy effects of a high-fat diet. The overall evidence seems to indicate that combined strategies based on exercise and dietary management can derive maximal benefit for neural health promotion. Furthermore, Pivik et al. (2012) recently examined the influence of a morning meal on complex mental functions in children (8–11 y). Brain activity was measured with electroencephalography (EEG) while children solved simple addition problems after an overnight fast and again after having either eaten or skipped breakfast. The fed children showed a significant increase in correct responses while children who continued to fast did not. The findings suggest that neural network activity involved in processing numerical information is functionally enhanced and performance is improved in children who have eaten breakfast, whereas greater mental effort is required for this mathematical thinking in children who skip breakfast (Pivik et al., 2012).
The Elderly - Preventing a Decline in Brain Function
Evidence from cross-sectional studies has consistently shown linear age-related declines in cognitive functions such as processing speed, short-term memory, working memory and long-term memory. The age-related decrements in cognition have been associated with changes in brain structure and function, and physical activity might play a central role in reducing age-associated cognitive losses (Hillman et al., 2006). Recent meta-analyses on the effects of physical activity on human cognitive aging showed that aerobic exercise had general and selective effects that were beneficial to cognitive function in older adults (Colcombe & Kramer, 2003; Etnier et al., 2006). These findings suggest that although cognitive performance declines in a global and linear fashion with age, physical activity and aerobic fitness may serve to protect against age-related loss of cognitive function, with the greatest benefits reported for processes requiring extensive amounts of executive control. There is a growing body of evidence to suggest that specific nutrients such as flavonoids and other polyphenols may also be capable of counteracting age-related neuronal and cognitive decline (Colcombe & Kramer, 2003; Gómez-Pinilla, 2011).
Brain Structure
It seems that these findings also translate into structural changes in the brain. A recent study showed that the volume of the hippocampus, a brain area very important in learning and memory, was greater in physically fit children when compared to age matched non-physically fit children (Chaddock et al., 2010a). Another study reported several additional brain regions were structurally different based on the child’s level of physical fitness. The dorsal striatum, believed to play a role in cognitive control and inhibition, was larger in children who were more physically fit (Chaddock et al., 2010b). It is possible that the observed structural differences between physically fit and unfit children may partly underlie the foundation for the functional brain differences seen in obese vs. healthy weight children.
Brain morphology will respond to specific stimuli throughout the lifespan. Raji et al. (2010) used functional magnetic resonance imaging (MRI) to assess gray and white matter volume atrophy in 94 elderly adults (mean age of 77 y). Results indicated that body mass index (BMI), fasting plasma insulin and type 2 diabetes were strongly associated with atrophy of the frontal, temporal and subcortical regions of the brain. These data indicated that being overweight or obese may be associated with marked decreases in brain volume and provide a greater understanding of the underlying causes of obesity-related changes in cognitive dysfunction. Given that several of the brain regions demonstrating decreases in brain volume are associated with attention, memory and the control of cognition, obesity-related deficits in cognitive and scholastic performance mediated by these brain regions might be expected.
It also appears that exercise training in older people can still influence brain morphology. Hippocampal and medial temporal lobe volumes were larger in higher-fit adults and physical activity training increased hippocampal perfusion. Erickson et al. (2011) clearly demonstrated, in a randomized controlled trial with 120 older adults (55-80 y), that one year of aerobic exercise training increased the size of the anterior hippocampus, leading to improvements in spatial memory. Exercise training increased hippocampal volume by 2%, effectively reversing the age-related loss in volume by one to two years. Hippocampal volume declined in the control group. Caudate nucleus and thalamus volumes were unaffected by the intervention. These findings indicated that aerobic exercise training was effective at reversing hippocampal volume loss in late adulthood, which is accompanied by improved memory function (Erickson et al., 2011).
Mechanisms for Exercise Benefits in the Brain
Animal research has shown that enriched environments, including access to running wheels, has a positive effect on neuronal growth and on the neural systems that are involved in learning and memory. This “neuroplasticity” refers to the ability of the brain to adapt to environmental change, respond to injury and acquire novel information by modifying neural connectivity and function. Compounds that promote neuroplasticity are called “neurotrophins” and they are capable of signaling neurons to survive, differentiate or grow. Neurotrophic factors not only play a role in neurobiology, but also in central and peripheral energy metabolism (Knaepen et al., 2010). Their effect on synaptic plasticity in the central nervous system (CNS) involves elements of cellular energy metabolism. Acute exercise and training seem to be key interventions to trigger the processes through which neurotrophins mediate energy metabolism and neural plasticity. Of all neurotrophins, brain-derived neurotrophic factor (BDNF) seems to be the most susceptible to regulation by exercise and physical activity (Knaepen et al., 2010). BDNF has a wide repertoire of neurotrophic and neuroprotective properties in the CNS and the periphery, namely: neuronal protection and survival; neurite expression; axonal and dendritic growth and remodeling; neuronal differentiation and synaptic plasticity such as synaptogenesis in arborizing axon terminals; and synaptic transmission efficacy. Animal studies also revealed a neuroendocrine and/or metabotrophic capacity of BDNF in the periphery. BDNF reduces food intake, increases oxidation of glucose, lowers blood glucose levels and increases insulin sensitivity. In animals, a high-fat diet reduces the hippocampal concentration of BDNF, but exercise is able to reverse this dietary decrease (Molteni et al., 2004). Furthermore, in mice it has been shown that there is a central interaction between the adipocyte-derived hormone leptin, which plays a key role in regulating appetite and energy metabolism, and BDNF expression in the hypothalamus (Komori et al., 2006). Araya et al. (2008) found that serum BDNF increases in insulin-resistant, overweight and obese subjects after a reduced-calorie diet. These findings confirm that BDNF is not only essential in the neuronal system, but is also intimately connected with central and peripheral molecular processes of energy metabolism and homeostasis (Wisse & Schwartz, 2003).
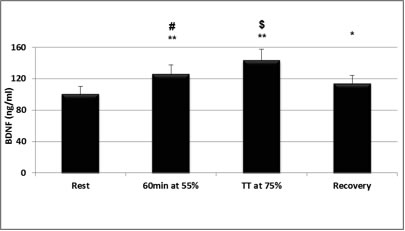
In search of mechanisms underlying plasticity and brain health, exercise is known to induce a cascade of molecular and cellular processes that support (brain) plasticity. BDNF could play a crucial role in these mechanisms. Therefore, since the early 1990s, studies started to investigate the effects of physical activity, acute exercise and/or training on the concentration of BDNF, first in animals (Radak et al., 2006; van Praag, 2009) and then in humans (Gold et al., 2003; Knaepen et al., 2010) (Figure 1). Little is known about the effect of resistance exercise on hippocampus-dependent memory, although this type of exercise is increasingly recommended to improve muscle strength and bone density and to prevent age-related disabilities. Resistance exercise increases cognitive performance especially in the elderly population. To explore the possible underlying mechanisms, Cassillhas et al. (2012) performed a study in which animals underwent no training or an aerobic training program or a resistance training program for eight weeks. After this period, both groups of trained animals showed increased cognition compared to control animals on the water maze test. However, different mechanisms were associated with the cognitive improvements in the two training groups. In endurance-trained rats, there was a significant increase in brain BDNF as well as its receptor, while in the resistance-trained animals BDNF did not increase. In the latter group, however, there was a significant increase of brain insulin-like growth factor (IGF-1), a molecule that stimulates nerve growth and its receptor. Further analysis showed that both training regimens induced an increase in the expression of synapsin 1 and synatophysin in the brain. Both molecules are important in the cascade necessary for nerve growth and therefore support the hypothesis that both aerobic and resistance exercise can employ divergent molecular mechanisms, but achieve similar results on learning and spatial memory.
NUTRITION TO INFLUENCE CENTRAL NERVOUS SYSTEM FATIGUE
Nutritional interventions can be used not only to influence cognition, but to prolong exercise performance or postpone fatigue. Fatigue can be defined as an acute impairment of exercise performance that leads to an inability to produce maximal force (Meeusen et al., 2006). It includes both an increase in the perceived effort necessary to exert a desired force or power output, and the eventual inability to produce that force or power output (Davis & Bailey, 1997). Fatigue may occur at the peripheral level and/or the level of the CNS.
Brain neurotransmitters and especially the central monoamines are strong candidates for inducing the centrally mediated effects of fatigue during exercise. The monoamines serotonin (5-HT), dopamine (DA) and noradrenaline (NA) play a key role in signal transduction between neurons, and exercise-induced changes in the concentrations of these neurotransmitters (especially 5-HT & DA) have been linked to central fatigue. Initially proposed by Acworth et al. (1986), Newsholme and co-workers (1987) developed the first hypothesis compromising changes in central neurotransmission to explain fatigue, i.e., the “Central Fatigue Hypothesis.” This hypothesis was based on disturbances in brain 5-HT concentrations, as this neurotransmitter is involved in changes in sleep-wakefulness, emotion, sleep, appetite, the hypothalamic-pituitary axis and numerous physiological functions (Meeusen et al., 2006). During exercise, the entry of tryptophan (the precursor of 5-HT) into the central nervous system through the blood-brain-barrier is favored by increased muscle use of branched-chain amino acids (BCAA) and elevated plasma fatty acids as this elevates the ratio of unbound tryptophan to BCAA. This increases the amount of tryptophan crossing the blood-brain barrier, consequently leading to higher 5-HT concentrations in the brain (Davis et al., 2000; Meeusen et al., 2006; Roelands & Meeusen, 2010). Events arising entirely from within the brain can influence an individual’s sensation of fatigue and thus potentially affect performance. This opens an opportunity to manipulate the central nervous system through changes in diet or supplementation with specific nutrients, including amino acids (BCAA, tyrosine), carbohydrates (CHO) and caffeine.
Branched Chain Amino Acids
Supplementation of BCAA has been proposed as a possible strategy to limit the development of central fatigue. Although this is a very attractive theory, there is limited or only circumstantial evidence to suggest that exercise performance in humans can be altered by nutritional manipulation through BCAA supplements, and today there is no convincing evidence that supplementation of BCAA will postpone fatigue during long-duration exercise (Meeusen & Watson., 2007).
Tyrosine
Tyrosine (hydroxyphenylalanine) can be synthesized in the body from phenylalanine, and is found in many high-protein foods such as soy products, chicken, turkey, fish, peanuts, almonds, avocados, milk, cheese, yogurt and sesame seeds. Acute consumption of tyrosine increases the ratio of tyrosine to other large neutral amino acids such as leucine, isoleucine, valine and tryptophan. A series of pre-clinical animal studies have been conducted that clearly indicate that tyrosine reduces many of the adverse effects of acute stress on cognitive performance in a wide variety of stressful environments. Although it has been difficult to conclusively demonstrate that tyrosine has beneficial effects in humans, in part due to ethical concerns, the preponderance of evidence suggests that tyrosine may be useful as an acute treatment to prevent stress-related declines in cognitive function. This may be especially so when exercising in the heat, as this represents a special demand on brain DA that is not apparent in temperate conditions (Roelands et al., 2008; Watson et al., 2005). Therefore, the brain tyrosine requirement may be greater with the cumulative demands of exercise and heat stress, and may become limiting to DA synthesis and release. Very recently, Tumilty et al. (2011) assessed the effects of acute tyrosine supplementation (150 mg/kg body mass) on exercise capacity in the heat. Eight healthy male subjects cycled until exhaustion at 68 ± 5% VO2peak. This study indicated, for the first time, that supplementing with a nutritional dopamine precursor one hour pre-exercise was associated with an increased exercise capacity in the heat. It appears that tyrosine availability, at least in part, may influence prolonged exercise tolerance with heat stress (Tumilty et al., 2011). Further studies are needed to identify the influence of regular supplementation of larger amounts of tyrosine (5-10 g) on health due to chronic changes in sympathetic nervous system activity.
Carbohydrate
While many studies have demonstrated an exercise-enhancing effect of CHO supplementation on peripheral metabolism, more recent evidence suggests that CHO feeding may also positively improve exercise performance by affecting the brain, either by a direct effect of glucose delivery to the brain or via neural signals sent from CHO receptors in the oral cavity to the brain. Carter et al. (2004) reported a 3% increase in performance (PLA 61.37 min; CHO 59.57 min) following the rinsing of a maltodextrin solution around in the mouth before and during exercise. No solution was actually ingested during the protocol, suggesting that this performance benefit may have been mediated through direct communication between receptors present in the mouth and the brain. The concept of the CHO mouth rinse has been supported by work investigating brain activity following the ingestion of a bolus of glucose (Liu et al., 2000), and activation of several brain regions after rinsing CHO solutions within the mouth (Chambers et al., 2009). These studies highlight a marked increase in brain activation, occurring immediately after CHO enters the mouth, with a second spike in activity observed 10 minutes following ingestion, presumably occurring as the substrate enters the circulation. Other groups have also examined the effects of a CHO mouth rinse on performance. Pottier et al. (2010) found a performance improvement on a 60-minute time trial rinsing with a CHO-electrolyte solution, while Rollo et al. (2008, 2010, 2011) showed ergogenic effects on different time trials. Interestingly, most studies that found an effect were carried out in the fasted state. When a CHO mouth rinse was performed in the fed state, no effect on performance in a 45-minute (Whitham & McKinney, 2007) and 60-minute time trial was observed (Beelen et al., 2009). The authors suggested that oral perception of carbohydrates perhaps only plays a role when muscle and liver glycogen stores are reduced. This finding was, however, not replicated in a very recent study where a mouth rinse with a maltodextrin solution increased time until exhaustion in both a fed and fasted state in non-athletic male subjects (Fares & Kayser, 2011). These findings are very novel and suggest an interesting mechanism of action. Further investigation of CHO receptors in the mouth is certainly warranted.
PRACTICAL APPLICATIONS AND CONCLUSIONS
Nutrition provides building blocks for the brain. Cognitive performance (e.g., mathematical thinking, simple addition problems) is better in children after a breakfast compared to the fasted state. There is a growing body of evidence to suggest that specific nutrients such as flavonoids and other polyphenols may be capable of counteracting age-related neuronal and cognitive decline. Exercise training in elderly people increases the size of the hippocampus (an area of the brain that is important for memory). Exercise positively influences neurotrophic factors (such as BDNF) leading to better learning and memory. Today, there is no convincing evidence that ingesting branched chain amino acids during prolonged exercise can postpone “central” fatigue. The ergogenic effect of carbohydrates during exercise is also present when washing the mouth with a CHO solution.
Exercise and nutrition clearly are both powerful means to positively influence the brain. We are only at the start of exploring what really happens in the brain during exercise, but it is clear that physical activity and nutrition have health-enhancing effects on the brain. In the near future, nutritional interventions will also focus on brain activity during exercise.
REFERENCES
Acworth, I., J. Nicholass, B. Morgan, and E.A. Newsholme (1986). Effect of sustained exercise on concentrations of plasma aromatic and branched-chain amino acids and brain amines. Biochem. Biophys. Res. Commun. 137:149-153.
Araya AV, X. Orellana, and J. Espinoza (2008). Evaluation of the effect of caloric restriction on serum BDNF in overweight and obese subjects: preliminary evidences. Endocrine 33:300-304.
Beelen, M., J. Berghuis, B. Bonaparte, S.B. Ballak, A.E. Jeukendrup, and L.J. van Loon (2009). Carbohydrate mouth rinsing in the fed state: lack of enhancement of time-trial performance. Int. J. Sport Nutr. Exerc. Metab. 19:400–409.
Carter, J.M., A.E.Jeukendrup, and D.A. Jones (2004). The effect of carbohydrate mouth rinse on 1-h cycle time trial performance. Med. Sci. Sports Exerc. 36:2107-2111.
Cassilhas, R.C., K.S. Lee, J. Fernandes, M.G.M. Oliveira, S. Tufik, R. Meeusen and M.T. De Mello (2012). Spatial memory is improved by aerobic and resistance exercise through divergent molecular mechanisms. Neuroscience 202:309–317
Chaddock, L., K.I. Erickson, and R.S. Prakash (2010a). A neuroimaging investigation of the association between aerobic fitness, hippocampal volume, and memory performance in preadolescent children. Brain Res. 1358:172–183.
Chaddock, L., K.I. Erickson, and R.S, Prakash (2010b). Basal ganglia volume is associated with aerobic fitness in preadolescent children. Dev. Neurosci. 32:249–256.
Chambers, E.S., M.W. Bridge, and D.A. Jones (2009). Carbohydrate sensing in the human mouth: effects on exercise performance and brain activity. J. Physiol. 587:1779-1794.
Colcombe, S. and A.F. Kramer (2003). Fitness effects on the cognitive function of older adults: a meta-analytic study. Psychol. Sci. 14:125–130.
Davis, C., and S. Cooper (2011). Fitness, fatness, cognition, behavior, and academic achievement among overweight children: Do cross-sectional associations correspond to exercise trial outcomes? Prev. Med. 52:S65–S69.
Davis, J.M., N.L. Alderson, and R.S. Welsh (2000). Serotonin and central nervous system fatigue: nutritional considerations. Am J. Clin. Nutr. 72:573S-578S.
Davis, J.M., and S.P. Bailey (1997). Possible mechanisms of central nervous system fatigue during exercise. Med. Sci. Sports Exerc. 29:45-57.
Erickson K, M. Voss, R. Prakash, C. Basak, A. Szabo, L. Chaddock, J. Kim, S. Heo, H. Alves, S. White, E. Wojcicki, E. Mailey, V. Vieira, S. Martin, B. Pence J. Woods E. McAuley and A. Kramer (2011). Exercise training increases the size of hippocampus and improves memory. PNAS 108:3017–3022.
Etnier, J. L. Nowell, P.M. Landers, D.M. and B. Sibley (2006). A meta-regression to examine the relationship between aerobic fitness and cognitive performance. Brain Res. Rev. 52:119–130.
Fares, E.J., and B. Kayser (2011). Carbohydrate mouth rinse effects on exercise capacity in pre- and postprandial states. J. Nutr. Metab. 2011:385926.
Goekint, M., E. Heyman, B. Roelands, R. Njemini, I. Bautmans, T. Mets, and R. Meeusen (2008). No influence of noradrenaline manipulation on acute exercise-induced increase of brain-derived neurotrophic factor. Med. Sci. Sports Exerc. 40:1990-1996.
Gold SM, K. Schulz, S. Hartmann, M. Mladek, U.E.Lang, R. Hellweg, R. Reer, K.M. Braumann, and C. Heesen (2003). Basal serum levels and reactivity of nerve growth factor and brain-derived neurotrophic factor to standardized acute exercise in multiple sclerosis and controls, J Neuroimmunol. 183:99-105.
Gómez-Pinilla, F. (2011). The combined effects of exercise and foods in preventing neurological and cognitive disorders. Prevent. Med. 52:S75–S80.
Hillman, .C, R. Motl, M. Pontifex, D. Posthuma, J. Stubbe, D. Boomsma, and E, de Geus (2006). Physical activity and cognitive function in a cross-section of younger and older community-dwelling individuals. Health Psych. 25:678–687.
Hoyland, A., L. Dye, and C.L. Lawton (2009). A systematic review of the effect of breakfast on the cognitive performance of children and adolescents. Nutr. Res. Rev. 22:220–243.
Knaepen, K., M. Goekint, E. Heyman, and R. Meeusen (2010). Neuroplasticity - exercise-induced response of peripheral brain-derived neurotrophic factor; a systematic review of experimental studies in human subjects. Sports Med. 40:765-801.
Komori. T., Y. Morikawa, K. Nanjo, and E. Senba (2006). Induction of brain-derived neurotrophic factor by leptin in the ventromedial hypothalamus. Neuroscience 139:1107-1115.
Liu, Y., J.H. Gao, H.L. Liu, and P.T. Fox (2000). The temporal response of the brain after eating revealed by functional MRI. Nature 405:1058-1062.
Meeusen, R., and P. Watson (2007). Amino acids and the brain: do they play a role in “central fatigue”? Int. J. Sports Nutr. Exerc. Metab. 17:S37-S46.
Meeusen, R., P. Watson, H. Hasegawa, B. Roelands, and M.F. Piacentini (2006). Central fatigue: the serotonin hypothesis and beyond. Sports Med. 36:881-909.
Molteni, R., A. Wu, and S. Vaynman (2004). Exercise reverses the harmful effects of consumption of a high-fat diet on synaptic and behavioral plasticity associated to the action of brain-derived neurotrophic factor. Neuroscience 123:429-440.
Newsholme, E.A., I. Acworth, and E. Blomstrand (1987). Amino acids, brain neurotransmitters and a function link between muscle and brain that is important in sustained exercise. In: G. Benzi (ed) Advances in Myochemistry. London: John Libbey Eurotext, pp. 127-133.
Parrott, M., and C. Greenwood (2007). Dietary influences on cognitive function with aging from high-fat diets to healthful eating Ann. N.Y. Acad. Sci. 1114:389–397.
Pivik, R., K. Tennal, S. Chapman, and Y. Gu (2012). Eating breakfast enhances the efficiency of neural networks engaged during mental arithmetic in school-aged children. Physiol. Behav.106:548–555.
Pottier, A., J. Bouckaert, W. Gilis, T. Roels, and W. Derave (2010). Mouth rinse but not ingestion of a carbohydrate solution improves 1 h cycle time trial performance. Scan. J. Med. Sci. Sports 20:105-111.
Radak, Z., A. Toldy, Z. Szabo, S. Siamilis, C. Nyakas, G. Silye, J. Jakus, and S. Goto (2006). The effects of training and detraining on memory, neurotrophins and oxidative stress markers in rat brain. Neurochem. Int. 49:387-392.
Raji, C.A., A.J. Ho, N.N. Parikshak, J.T. Becker, O.L. Lopez, L.H. Kuller, X. Hua, A.D. Leow, A.W. Toga, and P.M. Thompson (2010). Brain structure and obesity. Hum. Brain Mapp. 31:353–364.
Roelands, B., H. Hasegawa, P. Watson, M.F. Piacentini, L. Buyse, G. De Schutter, and R. Meeusen (2008). The effects of acute dopamine reuptake inhibition on performance. Med. Sci. Sports Exerc. 40: 879-858.
Roelands, B., and R. Meeusen (2010). Alterations in central fatigue by pharmacological manipulations of neurotransmitters in normal and high ambient temperature. Sports Med. 40:229-246.
Rollo, I., M. Cole, R. Miller, and C. Williams (2010). Influence of mouth rinsing a carbohydrate solution on 1-h running performance. Med. Sci. Sports Exerc. 42:798-804.
Rollo, I., C. Williams, N. Gant, and M. Nute (2008). The influence of carbohydrate mouth rinse on self-selected speeds during a 30-min treadmill run. Int. J. Sport. Nutr. Exerc. Metab. 18:585-600.
Rollo, I., C. Williams, and M. Nevill (2011). Influence of ingesting versus mouth rinsing a carbohydrate solution during a 1-h run. Med. Sci. Sports Exerc. 43:468-475.
Taki, Y., H. Hashizume, Y. Sassa, H. Takeuchi, M. Asano, K. Asano, and R. Kawashima (2010). Breakfast staple types affect brain gray matter volume and cognitive function in healthy children. PLoS ONE 5(12): e15213.
Tumilty, L., G. Davison, M. Beckmann, and R. Thatcher (2011). Oral tyrosine supplementation improves exercise capacity in the heat. Eur. J. Appl. Physiol. 111:2941-2950.
van Praag, H. ( 2009). Exercise and the brain: something to chew on. Trends Neurosci. 32:283-290.
Watson, P., H. Hasegawa, B. Roelands, M.F. Piacentini, R. Looverie, and R. Meeusen (2005). Acute dopamine/noradrenaline reuptake inhibition enhances human exercise performance in warm, but not temperate conditions. J. Physiol. 565:873–883.
Whitham, M., and J. McKinney (2007). Effect of a carbohydrate mouthwash on running time trial performance. J. Sports Sci. 25:1385-1392.
Wisse, B.E., and M. Schwartz (2003). The skinny on neurotrophins. Nat. Neurosci. 6:655-656.








