Key Terms
- Aging is accompanied by a decline in skeletal muscle mass and strength.
- The loss of muscle mass with aging is at least partly attributed to a blunted muscle protein synthetic response to food intake.
- Physical activity increases the sensitivity of skeletal muscle tissue to the anabolic properties of protein consumption.
- Resistance-type exercise training represents an effective means to attenuate age-related muscle loss and can be applied effectively to increase muscle mass, strength and functional performance in the older population.
- Dietary protein supplementation in the active older adult augments the gains in skeletal muscle mass and strength during prolonged resistance-type exercise training.
- Research is ongoing to define the optimal type and amount of protein and appropriate timing of protein supplementation to further augment the adaptive response to exercise training in the older population and support active aging.
Introduction
Aging is accompanied by the loss of skeletal muscle mass and strength. This loss of muscle mass and strength reduces functional capacity and increases the risk of injury and illness, including the development of chronic metabolic disease. The loss of muscle mass with aging has been attributed, at least partly, to a reduced muscle protein synthetic response to food intake, and this has been coined as “anabolic resistance.” Physical activity increases the sensitivity of skeletal muscle tissue to the anabolic properties of protein consumption and may compensate for the anabolic resistance of aging. In support, prolonged resistance-type exercise training can strongly augment muscle mass and strength in the older population. Dietary protein supplementation can be applied to further augment the adaptive response to a more active lifestyle, further increasing gains in skeletal muscle mass and strength. Recent work aims to define the optimal type and amount of dietary protein and appropriate timing of protein consumption to maximize the skeletal muscle adaptive response to physical activity in the older adult. This Sports Science Exchange article outlines the current understanding of the role of dietary protein intake to augment gains in skeletal muscle mass, strength and performance to support healthy, active aging.
Demographics show evidence of global aging with the number of individuals aged 60 years and over predicted to double by the year 2050 (WHO, 2015). Aging is accompanied by a progressive loss of skeletal muscle mass and strength, which reduces functional capacity and predisposes individuals to falls, fractures, metabolic disease and other health complications. The mechanisms responsible for the age-related loss of muscle mass and strength are complex and remain far from elucidated. However, reduced levels of physical activity, prevalence of disease, periods of bed rest following injury and/or hospitalization and an inadequate protein content in the diet all appear to contribute to the loss of muscle mass with aging. Older adults who maintain a higher level of physical activity throughout the lifespan generally experience far less muscle loss when compared with sedentary, age-matched controls (Shephard et al., 2013). Consequently, retaining an active lifestyle seems to be a prerequisite for healthy aging. However, even the active older adult experiences some level of muscle and strength loss with advancing age. A modest loss of muscle mass can compromise strength, oxidative capacity and overall functional performance, and may hinder the ability to maintain a healthy, active lifestyle. Consequently, age-related muscle loss should not be regarded solely as a challenge for clinically compromised elderly; it should also be a concern for healthy older adults trying to remain (or become more) physically active.
Regulation of Muscle Mass Maintenance
Skeletal muscle protein is constantly being synthesized and broken down, with a turnover rate of about 1-2% per day (Figure 1).
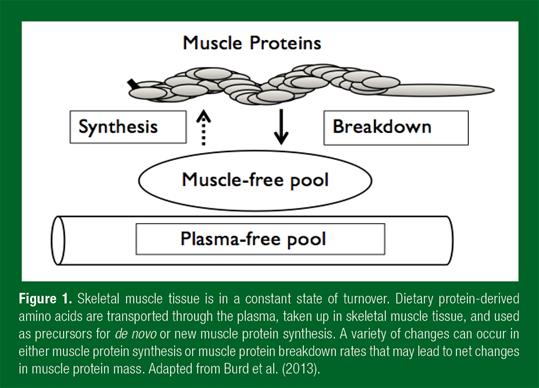
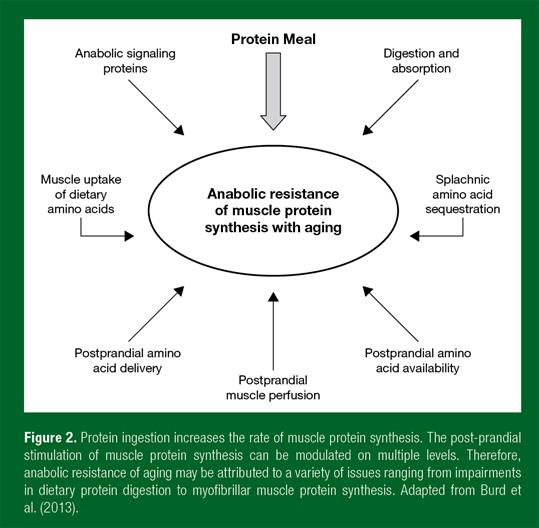
The rate of skeletal muscle protein synthesis is regulated by two main metabolic stimuli, food intake and physical activity. Food intake, or specifically protein ingestion, directly elevates muscle protein synthesis rates. The dietary protein-derived essential amino acids, and leucine in particular, act as signaling molecules activating anabolic pathways in skeletal muscle tissue and by providing precursors for muscle protein synthesis. Ingestion of a meal-like amount of dietary protein (~20 g) elevates muscle protein synthesis rates for several hours following ingestion, resulting in net muscle protein accretion. The post-prandial anabolic response to meal ingestion compensates for muscle protein loss in the post-absorptive fasting state, thereby allowing muscle mass maintenance. Recent work suggests that the muscle protein synthetic response to food intake is blunted in older vs. younger adults. This reduced responsiveness to meal ingestion in older adults, or “anabolic resistance,” is now commonly believed to represent a key factor in the loss of muscle mass – the development of sarcopenia (Burd et al., 2013). However, the exact mechanisms underlying anabolic resistance remain to be established. Impairments in the anabolic response to feeding may reside at the level of protein digestion, amino acid absorption, the post-prandial hormonal response and subsequent microvascular perfusion, amino acid uptake in skeletal muscle tissue, intramuscular signaling and/or subsequent myofibrillar muscle protein synthesis (Figure 2).
Interestingly, physical activity makes skeletal muscle tissue more sensitive to the anabolic properties of amino acid or protein administration (Burd et al., 2011). Physical activity performed prior to the ingestion of a meal-like amount of dietary protein can compensate for anabolic resistance in the older adult, allowing more of the ingested protein to be used for de novo muscle protein synthesis (Pennings et al., 2011a). These findings suggest that anabolic resistance in the older adult may be, at least partly, attributed to differences in physical activity levels as opposed to aging per se. In support, we (Wall et al., 2013b) as well as others (Glover et al., 2008), have demonstrated that a short period of reduced physical activity can rapidly induce a state of anabolic resistance. This implies that older people may overcome anabolic resistance and attenuate age-related muscle loss by increasing their physical activity level. It is evident that we need to make use of the anabolic properties of both physical activity and nutrition to optimize post-prandial muscle protein accretion and facilitate muscle mass maintenance in the older population.
Timing of Dietary Protein Intake
Besides food intake, physical activity represents another key anabolic stimulus. A single bout of exercise increases muscle protein synthesis as well as protein breakdown rates, albeit the latter to a lesser extent, thereby improving muscle protein balance. However, in the absence of nutrient intake, muscle protein balance will remain negative. Protein ingestion following exercise further increases muscle protein synthesis rates and inhibits muscle protein breakdown, resulting in net muscle protein accretion. The ingestion of dietary protein in close temporal proximity to physical activity or exercise has a synergistic impact on muscle protein synthesis rates in young adults, such that greater muscle protein synthesis rates are observed when compared to settings where a single stimulus is provided (Biolo et al., 1997; Moore et al., 2005; 2009b; Pennings et al., 2011a). In addition to the acute effects of exercise on muscle protein synthesis, a single bout of resistance-type exercise increases the sensitivity of skeletal muscle tissue to the anabolic properties of amino acid or protein feeding for at least 24 h following cessation of exercise (Burd et al., 2011). This suggests that the older individual may also experience a greater rise in post-prandial muscle protein synthesis following meals consumed 1-2 d following a single bout of exercise.
Ingestion of a protein-rich meal generally increases muscle protein synthesis rates for ~4 h, with peak protein synthesis rates being reached ~2 h after food ingestion (Moore et al., 2009b). Consequently, athletes trying to gain muscle mass and strength are generally advised to consume 4-6 smaller, protein-dense meals per day (Moore et al., 2009a). Similar guidelines could be applied to the older adult when trying to maintain or increase muscle mass. Recent work from our laboratory has shown that it is also feasible to increase muscle protein synthesis rates during overnight sleep by protein administration prior to sleep (Groen et al., 2012). Consequently, a small protein-rich meal could be consumed prior to sleep. In short, consuming four protein-rich meals per day, each containing ample protein, will facilitate muscle mass maintenance and support greater gains in muscle mass during resistance-type exercise training.
Type and Amount of Dietary Protein
An active lifestyle and ample dietary protein consumption, distributed over various meals throughout the day, improves the capacity to maintain skeletal muscle tissue with aging. The muscle protein synthetic response to each individual meal is further determined by the type and amount of protein consumed with each meal. Various studies have assessed the type (Boirie et al., 1997; Pennings et al., 2013; Tang et al., 2009; West et al., 2011) and amount (Moore et al., 2009a; Pennings et al., 2012; Witard et al., 2014; Yang et al., 2012) of dietary protein that can maximize post-prandial muscle protein synthesis rates. The anabolic potential of a dietary protein source is largely determined by its digestion and absorption kinetics and amino acid composition. A more rapidly digestible protein source, such as whey protein, can elicit a greater muscle protein synthetic response when compared with more slowly digestible protein sources such as casein (Pennings et al., 2011b; Tang et al., 2009). Even when digestion and absorption kinetics are similar, differences in the post-prandial muscle protein synthetic response between various types of dietary protein can be demonstrated (Pennings et al., 2011b). The latter is likely attributed to the leucine content of the protein source, with the ingestion of a more leucine-rich protein source resulting in a greater postprandial muscle protein synthetic response (Pennings et al., 2011b; Wall et al., 2013a). In support, fortifying a single bolus of casein with 2-3 g of free leucine can strongly augment the post-prandial anabolic response (Katsanos et al., 2006; Wall et al., 2013a). Consequently, a rapidly digestible protein source with a high leucine content seems to be preferred to stimulate post-prandial muscle protein accretion.
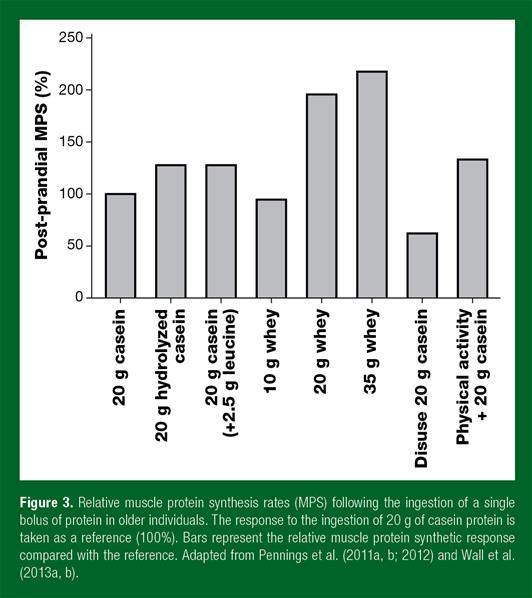
The muscle protein synthetic response following protein ingestion also increases in a dose-dependent manner (Moore et al., 2009a; Pennings et al., 2012; Witard et al., 2014; Yang et al., 2012), with maximal post-exercise muscle protein synthesis rates being achieved in young men following the ingestion of 20 g of egg or whey protein (Moore et al., 2009a, Witard et al., 2014, Yang et al., 2012). However, in elderly individuals, more protein may be required to maximize post-prandial muscle protein synthesis rates (Churchward-Venne et al., 2016). Ingestion of 35 g of whey protein has been shown to increase post-prandial muscle protein synthesis rates to a greater extent when compared with the ingestion of 20 g protein (Pennings et al., 2012). In agreement, ingestion of 40 g of whey protein during recovery from exercise has been shown to result in greater post-exercise muscle protein synthesis rates when compared with ingesting 20 g of whey protein in older men (Yang et al., 2012). Though many questions relating to the required amount of dietary protein to maximize post-prandial and/or post-exercise muscle protein synthesis remain, evidence thus far indicates that ingestion of 30-40 g of a high-quality dietary protein source immediately following exercise and at regular intervals thereafter may best support muscle reconditioning in the active older adult (Figure 3).
Under conditions where ample protein is provided following exercise, the type and composition of the protein source may be of less relevance due to the impact of the previous exercise bout on the sensitivity of skeletal muscle tissue to the anabolic properties of protein ingestion. Furthermore, as protein is generally consumed in a food matrix with co-ingestion of other macronutrients we can only speculate on the exact impact of the type and amount of dietary protein consumed on the post-prandial or post-exercise muscle protein synthetic response. Recently, work has started to address the impact of other macronutrients on the post-prandial muscle protein synthetic response (Gorissen et al., 2014; Hamer et al., 2013). It seems evident that frequent consumption of protein-dense meals containing 30-40 g of a high-quality protein source would be most effective to stimulate post-prandial muscle protein accretion in the older adult.
Protein Supplementation During Exercise Training
Prolonged resistance-type exercise training represents the most effective strategy to increase muscle mass and strength, as well as functional capacity in the elderly. There are no non-responders to the many benefits of resistance-type exercise training in older men or women (Churchward-Venne et al., 2015). An exercise intervention program fitted to the individual needs of the older adults can prevent or even reverse the loss of muscle mass and strength. Dietary protein consumption facilitates and supports the skeletal muscle adaptive response to a bout of resistance-type exercise and, as such, may improve exercise training efficiency. In agreement, various studies have confirmed the clinical benefits of dietary protein supplementation during prolonged resistance-type exercise training in the older population, with greater gains in muscle mass and strength being achieved following protein supplementation (Cermak et al., 2012). However, there are also many other studies that failed to confirm the proposed surplus benefits of protein supplementation. The apparent discrepancy in the literature is likely attributed to the use of relatively small subject cohorts and the application of study designs prescribing a less than optimal protein supplementation regimen. A recent meta-analysis thoroughly examined the literature investigating the proposed benefits of protein supplementation to further augment the adaptive response to prolonged resistance-type exercise training in young and older men (Cermak et al., 2012). Older individuals tended to achieve 30-40% greater gains in muscle mass and strength following protein supplementation during prolonged resistance-type exercise training (Cermak et al., 2012). Protein supplementation seems to be of even greater relevance in more clinically compromised pre-frail older adults, in whom additional protein seems a prerequisite to allow a net gain in muscle mass following 6 months of resistance-type exercise training (Tieland et al., 2012). These data illustrate the importance of dietary protein supplementation to support muscle reconditioning for the active older adult. It seems evident that we need to define and apply the most appropriate type, dose and timing of protein supplementation to maximize gains in muscle mass, strength and functional performance following prolonged exercise interventions in various healthy and more clinically compromised older subpopulations.
Nutritional Compounds to Support Active Aging
A healthy, well-balanced diet should form the basis of any nutritional plan aiming to improve muscle reconditioning. Beyond the timed consumption of sufficient high-quality protein, other nutritional compounds may be able to enhance the skeletal muscle adaptive response to exercise training in the older adults. Creatine supplementation has been widely used by recreational and competitive athletes to increase high-intensity exercise performance and/or to increase muscle mass and strength during prolonged resistance-type exercise training. Creatine (monohydrate) is generally supplemented in relatively large doses (four times 5 g/d) for a relatively short 5-7 d period to increase skeletal muscle phosphocreatine content. A maintenance dose of 2-5 g of creatine/d is subsequently taken to maintain the elevated muscle phosphocreatine levels. Ergogenic benefits have been attributed to the greater phosphocreatine stores allowing more adenosine triphosphate to be available for energy provision during intermittent high-intensity exercise, allowing a more intense workout and greater training adaptation. A recent meta-analysis concluded that creatine supplementation can augment gains in muscle mass and strength during a period of exercise training in older adults (Devries & Phillips, 2014). It could be speculated that this may be of more relevance to the experienced older athlete, having already maximized the exercise-induced gains in muscle mass and strength, as opposed to more novice older adults just starting an exercise intervention program.
Another nutritional compound that has recently gained much attention is the fish oil-derived omega-3 fatty acids. It has been reported that omega-3 supplementation increases the muscle protein synthetic response to amino acid administration (Smith et al., 2011). Recent work reported that fish oil supplementation (2 g/d) augmented gains in muscle mass, strength and functional capacity in older women following 3 months of resistance-type exercise training (Rodacki et al., 2012). More work is warranted to define fish oil supplementation as an effective adjuvant nutritional strategy to augment the skeletal muscle adaptive response to a more active lifestyle in the older population.
PRACTICAL RECOMMENDATIONS
The nutritional recommendations for active older adults include:
- Provide sufficient protein (at least 30 g) with each main meal
- Ensure sufficient protein intake with breakfast
- Ingest 30-40 g protein immediately after exercise
- Consume 30-40 g protein prior to sleep
Summary
Aging is associated with a blunted muscle protein synthetic response to food intake. This “anabolic resistance” is now believed to contribute to the age-related loss of muscle mass and strength. Physical activity and exercise re-sensitize skeletal muscle tissue to the anabolic properties of protein ingestion in older adults. Consuming dietary protein in close temporal proximity to physical activity increases muscle protein accretion and allows more of the ingested protein to be used for de novo muscle protein synthesis. Consuming four protein-dense meals per day may optimize post-prandial muscle protein accretion and may facilitate the gains in muscle mass and strength observed during prolonged exercise training. A 30-40 g dose of a high-quality protein likely represents a proper amount to optimize (post-exercise) muscle protein synthesis rates in the older adult. Post-prandial muscle protein synthesis rates may be further improved by consuming those dietary proteins that are rapidly digested and absorbed and are rich in leucine. Dietary protein supplementation during prolonged resistance-type exercise training augments the skeletal muscle adaptive response, resulting in greater gains in skeletal muscle mass and strength in both young and older adults. The use of nutritional compounds such as creatine (monohydrate) and fish oil-derived fatty acids may further enhance the benefits of prolonged exercise training in the older ath-lete. More work will be directed to the development of nutritional strategies that facilitate the skeletal muscle adaptive response to exercise training in the older population, thereby contributing to more healthy active aging.
References
Biolo, G., K. Tipton, S. Klein, and R. Wolfe (1997). An abundant supply of amino acids enhances the metabolic effect of exercise on muscle protein. Am. J. Physiol. 273:E122-E129.
Boirie, Y., M. Dangin, P. Gachon, M. Vasson, J. Maubois, and B. Beaufrère (1997). Slow and fast dietary proteins differently modulate postprandial protein accretion. Proc. Natl. Acad. Sci. USA. 94:14930-14935.
Burd, N., S. Gorissen, and L. van Loon (2013). Anabolic resistance of muscle protein synthesis with aging. Exerc. Sport Sci. Rev. 41:169-173.
Burd, N., D. West, D. Moore, P. Atherton, A. Staples, T. Prior, J. Tang, M. Rennie, S. Baker, and S. Phillips (2011). Enhanced amino acid sensitivity of myofibrillar protein synthesis persists for up to 24 h after resistance exercise in young men. J. Nutr. 141:568-573.
Churchward-Venne, T., A. Holwerda, S. Phillips, and L. van Loon (2016). What is the optimal amount of protein to support post-exercise skeletal muscle reconditioning in the older adult? Sports Med. E-pub ahead of print. PMID 26894275.
Churchward-Venne, T., M. Tieland, L. Verdijk, M. Leenders, M. Dirks, L. de Groot, and L van Loon (2015). There are no nonresponders to resistance-type exercise training in older men and women. J. Am. Med. Dir. Assoc. 16:400-411.
Cermak, N., P. Res, L. de Groot, W. Saris, and L. van Loon (2012). Protein supplementation augments the adaptive response of skeletal muscle to resistance-type exercise training: a meta-analysis. Am. J. Clin. Nutr. 96:1454-1464.
Devries, M.C., and S. Phillips (2014). Creatine Supplementation during Resistance Training in Older Adults-a Meta-analysis. Med. Sci. Sports Exerc. 46:1194-1203.
Glover, E., S. Phillips, B. Oates, J. Tang, M. Tarnopolsky, A. Selby, K. Smith, and M. Rennie (2008). Immobilization induces anabolic resistance in human myofibrillar protein synthesis with low and high dose amino acid infusion. J. Physiol. 586:6049-6061.
Gorissen, S., N. Burd, H.M. Hamer, A. Gijsen, B. Groen, and L. van Loon (2014). Carbohydrate co-ingestion delays dietary protein digestion and absorption but does not modulate postprandial muscle protein accretion. J. Clin. Endocrinol. Metab. 99:2250-225
Groen, B., P. Res, B. Pennings, E. Hertle, J. Senden, W. Saris, and L. van Loon (2012). Intragastric protein administration stimulates overnight muscle protein synthesis in elderly men. Am. J. Physiol. 302:E52-E60.
Hamer, H., B. Wall, A. Kiskini, A. de Lange, B. Groen, J. Bakker, A. Gijsen, L. Verdijk, and L. van Loon (2013). Carbohydrate co-ingestion with protein does not further augment post-prandial muscle protein accretion in older men. Nutr. Metab. 10:10-15.
Katsanos, C., H. Kobayashi, M. Sheffield-Moore, A. Aarsland, and R. Wolfe (2006). A high proportion of leucine is required for optimal stimulation of the rate of muscle protein synthesis by essential amino acids in the elderly. Am. J. Physiol. 291:E381-E387.
Moore, D., S. Phillips, J. Babraj, K. Smith, and M. Rennie (2005). Myofibrillar and collagen protein synthesis in human skeletal muscle in young men after maximal shortening and lengthening contractions. Am. J. Physiol. 288:E1153-E1159.
Moore, D., M. Robinson, J. Fry, J. Tang, E. Glover, S. Wilkinson, T. Prior, M. Tarnopolsky, and S. Phillips (2009a). Ingested protein dose response of muscle and albumin protein synthesis after resistance exercise in young men. Am. J. Clin. Nutr. 89: 161-168.
Moore, D., J. Tang, N. Burd, T. Rerecich, M. Tarnopolsky, and S. Phillips (2009b). Differential stimulation of myofibrillar and sarcoplasmic protein synthesis with protein ingestion at rest and after resistance exercise. J. Physiol. 587:897-904.
Pennings, B., R. Koopman, M. Beelen, J. Senden, W. Saris, and L. van Loon (2011a). Exercising before protein intake allows for greater use of dietary protein-derived amino acids for de novo muscle protein synthesis in both young and elderly men. Am. J. Clin. Nutr. 93:322-331.
Pennings, B., Y. Boirie, J. Senden, A. Gijsen, H. Kuipers, and L. van Loon (2011b). Whey protein stimulates postprandial muscle protein accretion more effectively than do casein and casein hydrolysate in older men. Am. J. Clin. Nutr. 93:997-1005.
Pennings, B., B. Groen, A. de Lange, A. Gijsen, A. Zorenc, J. Senden, and L. van Loon (2012). Amino acid absorption and subsequent muscle protein accretion following graded intakes of whey protein in elderly men. Am. J. Physiol. 302:E992-E999.
Pennings, B., B. Groen, J. van Dijk, A. de Lange, A. Kiskini, M. Kuklinski, J. Senden, and L. van Loon (2013). Minced beef is more rapidly digested and absorbed than beef steak, resulting in greater postprandial protein retention in older men. Am. J. Clin. Nutr. 98:121-128.
Rodacki, C., A. Rodacki, G. Pereira, K. Naliwaiko, I. Coelho, D. Pequito, and L. Fernandes (2012). Fish-oil supplementation enhances the effects of strength training in elderly women. Am. J. Clin. Nutr. 95:428-436.
Shephard R.J., H. Park, S. Park, and Y. Aoyagi (2013). Objectively measured physical activity and progressive loss of lean tissue in older Japanese adults: longitudinal data from the Nakanojo study. J. Am. Geriatr. Soc. 61:1887-1893.
Smith, G., P. Atherton, D. Reeds, B. Mohammed, D. Rankin, M. Rennie, and B. Mittendorfer (2011). Omega-3 polyunsaturated fatty acids augment the muscle protein anabolic response to hyperinsulinaemia-hyperaminoacidaemia in healthy young and middle-aged men and women. Clin. Sci. 121:267-278.
Tang, J., D. Moore, G. Kujbida, M. Tarnopolsky, and S. Phillips (2009). Ingestion of whey hydrolysate, casein, or soy protein isolate: effects on mixed muscle protein synthesis at rest and following resistance exercise in young men. J. Appl. Physiol. 107:987-992.
Tieland, M., M. Dirks, N. van der Zwaluw, L. Verdijk, O. van de Rest, L. de Groot, and L. van Loon (2012). Protein supplementation increases muscle mass gain during prolonged resistance-type exercise training in frail elderly people: a randomized, double-blind, placebo-controlled trial. J. Am. Med. Dir. Assoc. 13:713-719.
Wall, B., H. Hamer, A. de Lange, A. Kiskini, B. Groen, J. Senden, A. Gijsen, L. Verdijk, and L. van Loon (2013a). Leucine co-ingestion improves post-prandial muscle protein accretion in elderly men. Clin. Nutr. 32:412-419.
Wall, B., T. Snijders, J. Senden, C. Ottenbros, A. Gijsen, L. Verdijk and L. van Loon (2013b). Disuse impairs the muscle protein synthetic response to protein ingestion in healthy men. J. Clin. Endocrinol. Metab. 98:4872-4878.
West, D., N. Burd, V. Coffey, S. Baker, L. Burke, J. Hawley, D. Moore, T. Stellingwerff, and S. Phillips (2011). Rapid aminoacidemia enhances myofibrillar protein synthesis and anabolic intramuscular signaling responses after resistance exercise. Am. J. Clin. Nutr. 94:795-803.
WHO (2015). World report on ageing and health. http://www.who.int/ageing/events/world-report-2015-launch/en/
Witard, O., S. Jackman, L. Breen, K. Smith, A. Selby, and K. Tipton (2014). Myofibrillar muscle protein synthesis rates subsequent to a meal in response to increasing doses of whey protein at rest and after resistance exercise. Am. J. Clin. Nutr. 99:86-95.
Yang, Y., L. Breen, N. Burd, A. Hector, T. Churchward-Venne, A. Josse, M. Tarnopolsky, and S. Phillips (2012). Resistance exercise enhances myofibrillar protein synthesis with graded intakes of whey protein in older men. Br. J. Nutr. 7:1-9.